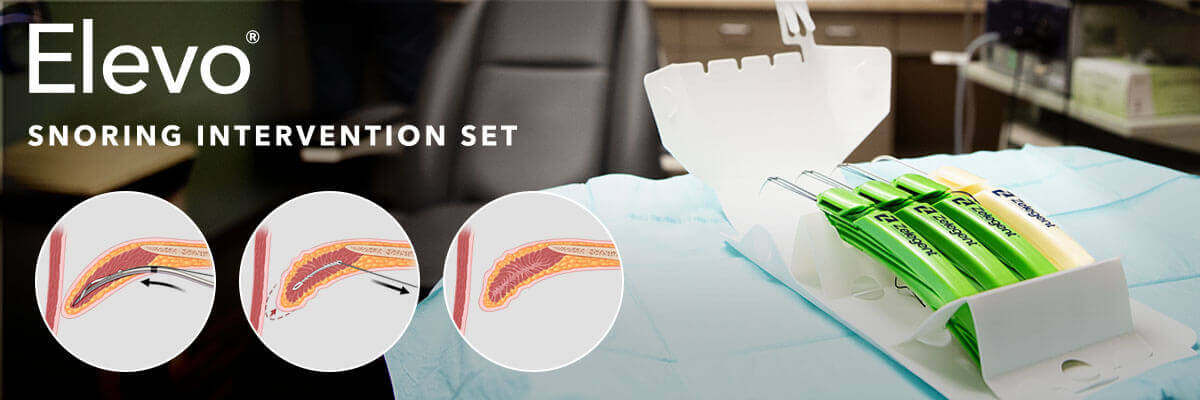
Elevate the palate. Alleviate snoring.1,2
What is the Elevo® Snoring Intervention Set?
Created by Zelegent, Inc., and distributed by Cook Medical, the Elevo Snoring Intervention Set is a specialized device designed to enhance patient treatment options for snoring. The concept of Elevo was developed by Dr. Yosef P. Krespi, who leads the New York Head and Neck Institute Sleep Disorder Center, and Dr. David O. Volpi, founder of the eos sleep network (formerly called the Manhattan Sleep Center). Their goal was to develop a minimally invasive device to be used by sleep specialty otolaryngologists to treat snoring at the palatal level without the need for invasive surgery.
How is the Elevo Snoring Intervention Set used?
The Elevo Snoring Intervention Set contains a delivery tool that allows the placement of bi-directional, barbed resorbable suture implants.1 Elevo is used in Elevoplasty®, a procedure that provides lift and stiffening to the soft palate. This procedure can be performed under local anesthesia in an office setting without the need for surgery. By providing the ability to both lift and stiffen the soft palate, Elevoplasty is the next step in the office-based treatment of primary snoring.
The Elevo Snoring Intervention Set is intended for use in stiffening the soft palate tissue, which may reduce the severity of snoring in some individuals. It is indicated for symptomatic, habitual, and social snoring due to palatal flutter.
Request a demo
Please submit the required fields to be connected with your local Cook representative. This form is intended for US-based physicians only.
By clicking ‘Submit,’ you agree to the terms and conditions for collecting and processing your personal information, as included in our customer data privacy notice.
Features and benefits
Effective procedure
After the lifting and stiffening action created in the soft palate, patients experienced a 30.3% reduction in sVAS at 180 days post-procedure.

Resorbable material
The Elevo suture implant is fully resorbable, minimizing the risk of extrusion that may occur with non-resorbable implants.1
Office-based approach
Elevoplasty provides a minimally invasive, office-based procedure for patients suffering from primary snoring.

Additional resources
- Zelegent, Inc. website
- S.I.LE.N.C.E clinical trial
- Elevo product webpage
- Cook Medical and Zelegent, Inc. begin sales and marketing agreement
- Cook Medical collaboration with Zelegent, Inc. receives FDA clearance for snoring product
- Elevo kit snoring intervention device. Premarket notification 510(k) K181107.
- Indications for Use: The Elevo Kit Snoring Intervention Device is intended for use in stiffening the soft palate tissue, which may reduce the severity of snoring in some individuals.
