Contact A Representative
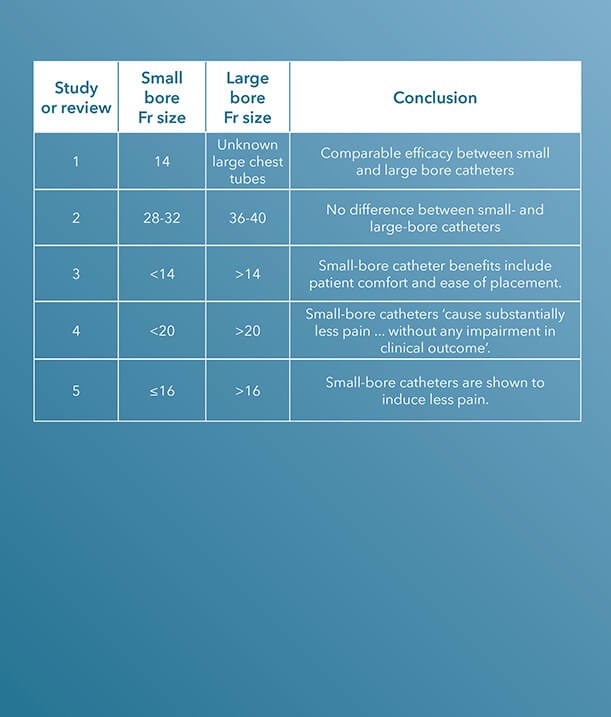
Since 1963, Cook Medical has been a champion of minimally invasive treatment options for patients. In the case of the centesis and drainage procedural areas, the healthcare industry discussion has largely centered around the efficacy of large-bore versus small-bore catheters.
Within the last decade, there have been several clinical studies and review articles published in peer-reviewed journals stating the increased efficacy and improvement of small-bore catheters versus large-bore catheters for pleural and pericardial drainage procedures.
To see Critical Care’s entire product portfolio of small-bore chest tubes for pleural and pericardial drainage, click here.
ARTICLE 1 (STUDY)
“Two-year experience of using pigtail catheters to treat traumatic pneumothorax: a changing trend,” published in The Journal of Trauma in 2011, is a study comparing small-bore, 14 Fr pigtail catheters to chest tubes in patients with nontraumatic pneumothoraces.
A chest tube, “because of its large caliber and significant trauma during an insertion, can cause pain, prevent full lung expansion, and worsen pulmonary outcome. Pigtail catheters (PC)… are smaller and less invasive,” the introduction to the study states.¹
The charts of 9,624 trauma patients over a two-year period at a Level I trauma center were evaluated. The study concludes that the “demographics, tube days, need for mechanical ventilation, and insertion-related complications were similar,” using these characteristics to determine that pigtail catheters have a comparable efficacy to chest tubes.1
ARTICLE 2 (STUDY)
36 to 40 Fr chest tubes were also compared to small-bore pigtail catheters in a similar study also published in The Journal of Trauma and Acute Care Surgery, titled “Does size matter? A prospective analysis of 28-32 versus 36-40 French chest tube size in trauma.”
There was no difference in the efficacy of drainage [or] rate of complications.2
In this study, the authors claim that “the optimal chest tube size for the drainage of traumatic hemothoraces and pneumothoraces is unknown.”2 This study was designed to compare the efficacy of small-bore versus large-bore drainage catheters in patients with thoracic trauma.
Data were gathered on 293 patients who required open chest tube drainage within 12 hours of admission to a Level I trauma center.
“Clinical demographic data and outcomes including efficacy of drainage, complications, retained hemothoraces, residual pneumothoraces, need for additional tube insertion, video-assisted thoracoscopy, and thoracotomy were collected and analyzed by tube size,” the authors indicate.2 Small-bore catheters, ranging from 28 to 32 Fr, were compared with large chest tubes, ranging from 36 to 40 Fr; a total of 353 catheters, 186 small and 167 large, were placed during the study.
The authors determined that “the need for tube reinsertion, image-guided drainage, video-assisted thoracoscopy, and thoracotomy” are the same when comparing small-bore to large-bore catheters.2
“For injured patients with chest trauma, chest tube size did not impact the clinically relevant outcomes tested. There was no difference in the efficacy of drainage, rate of complications including retained hemothorax, need for additional tube drainage, or invasive procedures,” the study concludes.2
ARTICLE 3 (REVIEW)
In 2013, a paper published in Clinics in Chest Medicine aimed to generalize the use of small-bore catheters for drainage procedures, including in pleural infection.
In “Straightening out chest tubes: what size, what type, and when,” the authors state that “small-bore tubes (<14 Fr) are effective for most pleural processes. Various types of pneumothorax and malignant and infected complicated pleural effusions have been successfully managed with small-bore chest tubes.”3
Abundant literature supports a paradigm shift towards the more routine use of small-bore chest tubes for managing pleural disease.3
The authors also state that the benefits of using small-bore drainage catheters include patient comfort and ease of catheter placement.3
The study concludes that “abundant literature supports a paradigm shift towards the more routine use of small-bore chest tubes for managing pleural disease.”3
ARTICLE 4
A study published in CHEST, titled “The relationship between chest tube size and clinical outcome in pleural infection,” aimed to determine the optimal choice of drainage catheter sizes for pleural infection.
This multicenter study enrolled 405 patients and evaluated “the combined frequency of death and surgery, and secondary outcomes,” specifically “hospital stay, change in chest radiograph, and lung function at 3 months,” in patients who received a variety of sizes of chest tubes. In 128 of the 405 patients, a pain scale was also included.4
“There was no significant difference in the frequency with which patients either died or required thoracic surgery in patients receiving chest tubes of varying sizes,” the study notes; however, “pain scores were substantially higher in patients receiving (mainly blunt dissection inserted) larger tubes.”4
The study concludes that “smaller, guide-wire-inserted chest tubes cause substantially less pain than blunt-dissection-inserted larger tubes, without any impairment in clinical outcome in the treatment of pleural infection.”4
ARTICLE 5 (REVIEW)
Although the precise optimal chest drain size remains unknown, a review article titled “Optimal chest drain size: the rise of the small-bore pleural catheter,” published in Seminars in Respiratory and Critical Care Medicine, asserts that “objective data supporting the use of large-bore [chest] tubes is scarce.”5
The onus now is on those who favor large tubes to produce clinical data to justify the more invasive approach.5
In support of the efficacy of small-bore drains, the article states, “Increasing evidence shows that small-bore catheters induce less pain and are of comparable efficacy to large-bore tubes, including in the management of pleural infection, malignant effusion, and pneumothoraces. The onus now is on those who favor large tubes to produce clinical data to justify the more invasive approach.”5
Interested in chatting with a Cook Medical representative?
Please submit the required information to connect with your local Cook representative. This form is intended for US-based physicians only. Please see our Privacy Statement for data protection notices relating to our collection and use of your data.
1. Kulvatunyou N, Vijayasekaran A, Hansen A, et al. Two-year experience of using pigtail catheters to treat traumatic pneumothorax: a changing trend.
J Trauma. 2011;71(5):1104-1107.
2. Inaba K, Lustenberger T, Recinos G, et al. Does size matter? A prospective analysis of 28-32 versus 36-40 French chest tube size in trauma. J Trauma Acute Care Surg. 2012;72(2):422-427.
3. Mahmood K, Wahidi MM. Straightening out chest tubes: what size, what type, and when. Clin Chest Med. 2013;34(1):63-71.
4. Rahman NM, Maskell NA, Davies CW, et al. The relationship between chest tube size and clinical outcome in pleural infection. Chest. 2010;137(3):536-543.
5. Fysh ET, Smith NA, Lee YC. Optimal chest drain size: the rise of small-bore pleural catheter. Semin Respir Crit Care Med. 2010;31(6):760-768.
Contact A Representative
We offer an extensive line of chest tubes for removing both air and fluid from the pleural and pericardial spaces. These products are available in a variety of designs and sizes, including several small-bore configurations, Seldinger and trocar options, and sets and trays to meet specific procedural preferences. View each product’s indications for use, specifications, components, and other resources by clicking the links below.
Pneumothorax catheters
 Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
• The enlarged sideports are designed to facilitate the drainage of air.
• The radiopaque catheter material enhances x-ray visualization.
• Pigtail catheters have a comparable efficacy to chest tubes in patients with pneumothorax.*
*Kulvatunyou N, Vijayasekaran A, Hansen A, et al. Two-year experience of using pigtail catheters to treat traumatic pneumothorax: a changing trend. J Trauma. 2011;71(5):1104–1107.
View more details about the Wayne Pneumothorax Catheter Set and Tray – Seldinger.
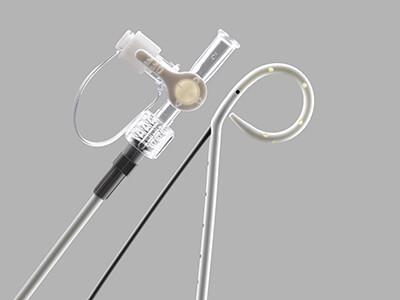 Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Features and benefits
• The needle obturator comes pre-assembled within the Wayne Pneumothorax Catheter to allow for direct-stick insertion.
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
• The enlarged sideports are designed to facilitate the drainage of air.
• The radiopaque catheter material enhances x-ray visualization.
• Pigtail catheters have a comparable efficacy to chest tubes in patients with pneumothorax.*
*Kulvatunyou N, Vijayasekaran A, Hansen A, et al. Two-year experience of using pigtail catheters to treat traumatic pneumothorax: a changing trend. J Trauma. 2011;71(5):1104–1107.
View more details about the Wayne Pneumothorax Catheter Set – Trocar.
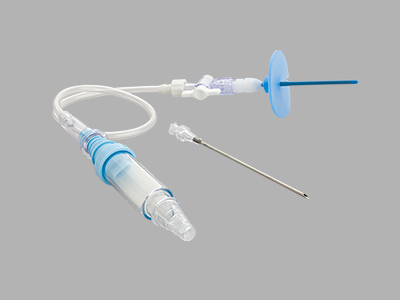 Used for emergency relief and temporary management of suspected tension pneumothorax.
Used for emergency relief and temporary management of suspected tension pneumothorax.
Features and benefits
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
View more details about the Cook Emergency Pneumothorax Set.
 Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Features and benefits
• The straight catheter is used for trocar placement.
• The radiopaque catheter material enhances x-ray visualization.
• The set and tray come with a vinyl connecting tube and a three-way stopcock.
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
View more details about the Pneumothorax Set and Tray.
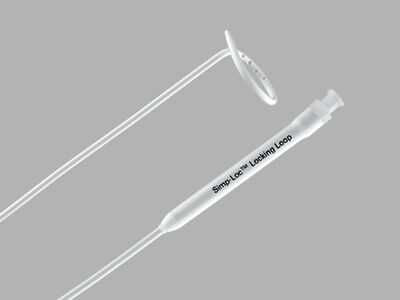 Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The 8.5 Fr catheter is designed for smaller patient anatomies.
• Sideports are positioned within the catheter pigtail and are designed to aid in drainage.
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
View more details about the Richli Pneumothorax Catheter Set.
 Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The radiopaque catheter material enhances x-ray visualization.
View more details about the Catheter Aspiration Set for Simple Pneumothorax.
Multipurpose catheters
 Used for the percutaneous introduction of a chest tube for pleural fluid drainage.
Used for the percutaneous introduction of a chest tube for pleural fluid drainage.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The centimeter-marked dilators allow for additional control over the insertion of a chest tube into the pleural space.
• The sideports are positioned at the distal end of the chest tube.
• The clear polyvinylchloride catheter with a radiopaque stripe allows fluid to be seen during the drainage procedure.
• The most proximal side hole is positioned on the radiopaque stripe to aid in confirming catheter positioning via x-ray.
• The double lumen set has an additional lumen available for aspiration or infusion.
• The Thal-Quick Chest Tube Adapter is sold separately.
View more details about the Thal-Quick Chest Tube Set and Tray.

After percutaneous insertion via the Seldinger technique, the pigtail catheter of the Fuhrman Pleural Drainage Set serves as a conduit to drain air or fluid from the pleural space to outside the body.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The catheter is available in a variety of sizes, ranging from 5 to 12 Fr, to cater to different patient anatomies.
• Sideports are positioned within the catheter pigtail and are designed to aid in drainage.
• Centimeter markings on the pigtail catheter guide insertion.
• The catheter material has radiopaque properties.
• Pigtail catheters have been shown to have a comparable efficacy to chest tubes in patients with pneumothorax.*
*Kulvatunyou N, Vijayasekaran A, Hansen A, et al. Two-year experience of using pigtail catheters to treat traumatic pneumothorax: a changing trend. J Trauma. 2011;71(5):1104–1107.
View more details about the Fuhrman Pleural/Pneumopericardial Drainage Set and Tray.
Pericardiocentesis catheters
 Used to remove fluid from the pericardial sac.
Used to remove fluid from the pericardial sac.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The 40 cm long catheter is designed to cater to larger patient anatomies.
• The radiopaque catheter material enhances x-ray visualization.
View more details about the Lock Pericardiocentesis Set and Tray.

Used to infuse or withdraw rinsing solutions in the peritoneal cavity.
Features and benefits
• Seldinger placement facilitates controlled, minimally invasive catheter introduction.
• The catheter is equipped with 90 sideports.
• The radiopaque catheter material enhances x-ray visualization.
View more details about the Peritoneal Lavage Set and Tray.
Accessories
 Used in conjunction with a pneumothorax aspiration catheter if incomplete expansion of the lung persists after mechanical aspiration of air.
Used in conjunction with a pneumothorax aspiration catheter if incomplete expansion of the lung persists after mechanical aspiration of air.
Features and benefits
• The set comes with the Cook Chest Drain Valve, which is designed to help remove air from the pleural cavity.
View more details about the Simple Pneumothorax Aspiration Accessory Set.
 Used for connection to other medical devices such as catheters.
Used for connection to other medical devices such as catheters.
Features and benefits
• The adapter is male Luer lock to a universal taper.
View more details about the Multipurpose Plastic Tubing Adapter.
 Used for the connection of an external drainage catheter to a drainage pouch.
Used for the connection of an external drainage catheter to a drainage pouch.
View more details about the Vinyl Connecting Tube.
 Used to provide separate sampling or an infusion port within Thal-Quick chest tubes.
Used to provide separate sampling or an infusion port within Thal-Quick chest tubes.
Features and benefits
• This accessory is compatible with the Thal-Quick Chest Tube Set and Tray.
View more details about the Thal-Quick Chest Tube Adapter.
Interested in chatting with a Cook Medical representative?
Please submit the required information to connect with your local Cook representative. This form is intended for US-based physicians only. Please see our Privacy Statement for data protection notices relating to our collection and use of your data.
Lung ultrasound is a necessary, key component in both pulmonary and critical care settings due to its high diagnostic accuracy and physicians’ ability to perform it at the bedside. In cases of pleural disease, lung ultrasound could be an essential component of care, from the initial diagnosis through clinical management and treatment.
Last year, Cook Medical had two physicians with expertise in the use of thoracic ultrasound for pleural disease share recent advances of this technique at two major industry conferences: the European Respiratory Society (ERS) International Congress and the CHEST Annual Meeting.
ERS International Congress
Luigi Vetrugno, MD, is a Professor of Anaesthesia and Intensive Care at the University Hospital of Udine in Udine, Italy. In his presentation, Thoracic ultrasound for pleural effusion, he discussed several studies he has published in recent years about the important role ultrasound plays in diagnosing and treating pleural effusion.
“Ultrasound plays a significant role in the education of physicians,” Dr. Vetrugno said. “They will need to be trained to view this technology as an extension of their senses, just as many generations have viewed the stethoscope in a similar way.”
In one study co-authored by Dr. Vetrugno and published in Critical Care Medicine, thoracic ultrasound is said to not only help physicians visualize pleural effusion, but also to help them distinguish between the different types that can be present.1
Additionally, “TUS [thoracic ultrasound] is essential during thoracentesis and chest tube drainage as it increases safety and decreases life-threatening complications. It is crucial not only during needle or tube drainage insertion, but also to monitor the volume of the drained PLEFF [pleural effusion].”1
An observational study co-authored by Dr. Vetrugno assessed “the prevalence of complications related to ultrasound-guided percutaneous small-bore pleural drain insertion.”2
He stated that “small-bore pleural drainage device insertion has become a first-line therapy for the treatment of pleural effusions.” In this study, ultrasound was used to assess the safety and complication rates in patients with pleural effusions. The study’s authors found ultrasound-guided placement to be a “safe procedure,” however, in the future, estimating the amount of pleural effusion by ultrasound will be necessary to standardize the procedure. The authors also concluded that for resident physicians “training and proficiency assessment should be formalized.”2
An additional article by Dr. Vetrugno in favor of ultrasound guidance can be found in Critical Ultrasound Journal, Respiratory and Pulmonary Medicine, and Annals of Intensive Care.
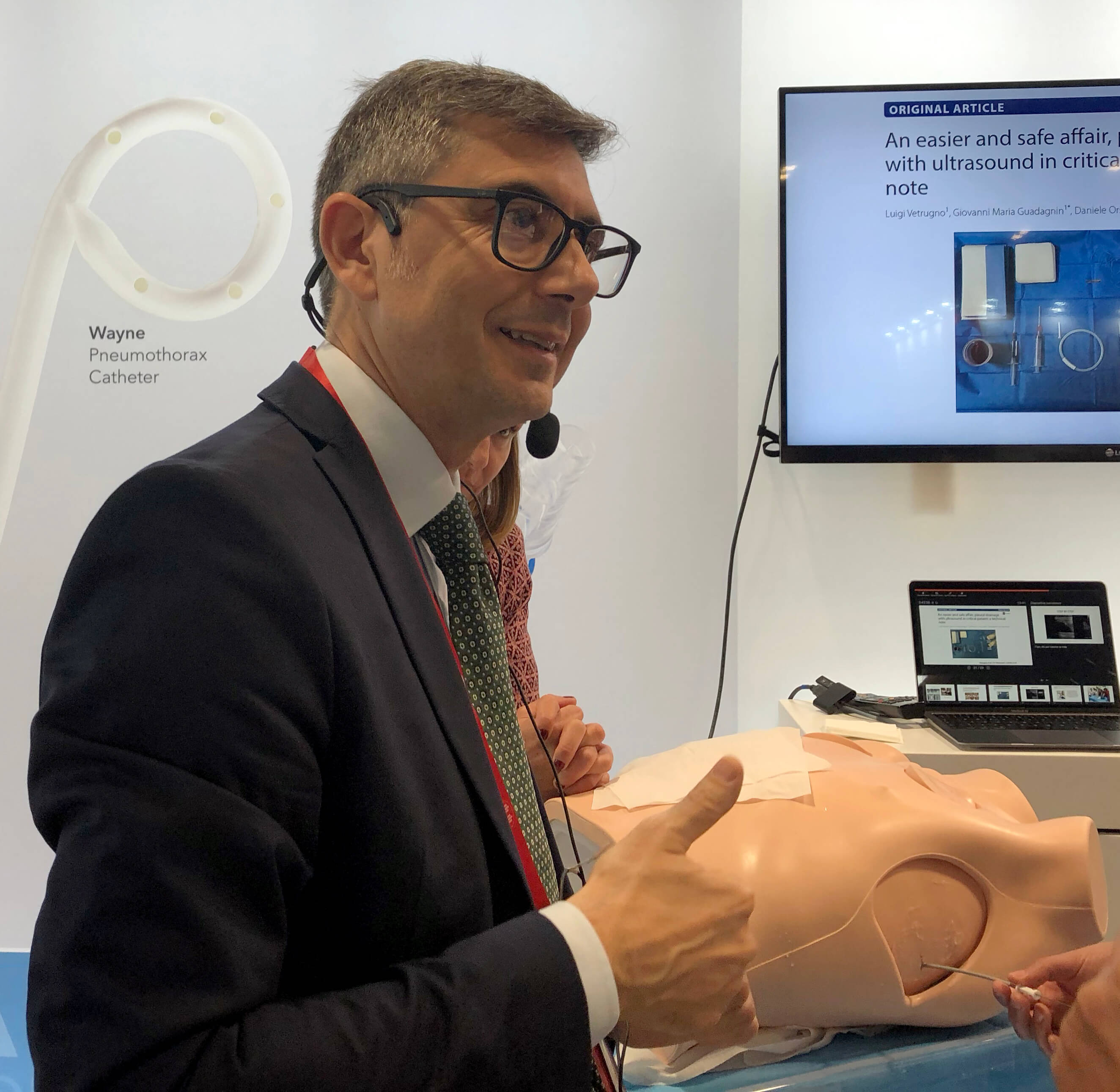 He has also submitted letters to the editor regarding the importance of patient position during ultrasound procedures.
He has also submitted letters to the editor regarding the importance of patient position during ultrasound procedures.
In one letter, Dr. Vetrugno advised for the patient to remain in supine position with a mild torso elevation of 15 degrees, not a semi-recumbent position with the torso at 40-45 degrees as previous authors stated.3 “This means that as fluid follow the law of gravity, an overestimation of the maximal distance between partial and visceral pleura could be obtained,” he said. This theory “overestimates in tall males with large thoracic circumference small effusions under 200 mL and in large ones above 1000 mL.”3
According to Dr. Vetrugno, this equation allows for a high mean prediction error, however, it is recognized that “an urgent standardization of the method to assess PLEFF [pleural effusion fluid] with lung ultrasound is needed to reach a definite conclusion.”3
In another letter to the editor, Dr. Vetrugno echoed his concerns that larger and more standardized clinical studies should be performed before a definite conclusion is reached.4
CHEST Annual Meeting
Seth Koenig, MD, FCCP, is the Director of Education and Professor of Medicine in the Division of Pulmonary Medicine at Montefiore Medical Center in New York City, New York. In his presentation, Ultrasonography for the diagnosis and management of pleural disease, he presented about the extensive benefits of using ultrasound, most notably regarding patient safety.
Dr. Koenig started his presentation with an important question, “How do we maximize the things we can do for our patients safely without using another service? And how does ultrasound help?”
To this question, the majority of the physician audience said that they routinely use ultrasound for general purposes, however, Dr. Koenig urged them to consider ultrasound for pleural effusions. “For me, I use ultrasound to do everything,” he said.
 Dr. Koenig explained that the use of ultrasound prevents a concept commonly known as “clinical and time dissociation.”
Dr. Koenig explained that the use of ultrasound prevents a concept commonly known as “clinical and time dissociation.”
“When you ask for another service, you invoke the ancient art of clinical and time dissociation,” he said. “For example, if you ask for a CAT scan, you will most likely have to involve a surgeon, an infectious disease doctor, and a pulmonologist. This can be confusing for the patient. Someone else reads the exam, you get the results of that exam, but they’re not intimately involved in the patient. This takes time for the exams to come in, plus, you have to move the patient. Is this what you do, or should we figure out a better way?”
Ultrasound, according to Dr. Koenig, not only provides added clarity in terms of a diagnostic approach, but it may also reduce the number of specialists involved in the diagnostic process.
“Every single patient that we see from the pulmonary and from critical care departments gets an ultrasound,” he said.
Additional advantages of ultrasound, according to Dr. Koenig, include:
- Short learning curve
- Portable; the patient does not need to be transferred
- Decrease the need for chest x-rays
- Vessels are visible; helps to avoid post-procedural bleed
- Can reduce likelihood of a visceral laceration, vascular injury, or pneumothorax
- Ability to monitor and record complications
Dr. Koenig also said that using ultrasound alleviates the need to precisely place a chest tube in the “triangle of safety,” a small, preferred site of insertion as determined by the British Thoracic Society.5
“The triangle isn’t a problem anymore,” he said. “As long you have a nice space, you can stick the needle wherever is convenient for you and the patient because you can see everything. As long as you know where you are and your wire guide is well placed, it doesn’t matter what you insert after. The point is, if you can see it, you can do something about it.”
Dr. Koenig ended his lecture by emphasizing the most important reason physicians should consider the use of ultrasound for pleural diseases: the patients.
“We have to think about our patients before we do things,” he said. “In 2019, I believe patients see too many doctors. They get a doctor for this, they get a doctor for that, what happens is, patients get really confused, and so if you can decrease the number of people and the number of procedures that a person has, they thank you for it, believe it or not. In conclusion, ultrasound is good for the patient, but it’s also good for you. Ultrasound helps with diagnostic and therapeutic planning. It helps to diagnose complications. It’s helpful to follow the progress of a pleural effusion, and it’s obviously extremely good for documenting post-procedure pneumothoraces.”

Dr. Vetrugno is not a paid consultant of Cook Medical.
Dr. Koenig is a paid consultant of Cook Medical.
1. Brogi E, Gargani L, Bignami E, et al. Thoracic ultrasound for pleural effusion in the intensive care unit: a narrative review from diagnosis to treatment. Crit Care. 2017;21:325.
2. Vetrugno L, Guadagnin GM, Barbariol F, et al. Assessment of pleural effusion and small pleural drain insertion by resident doctors in an intensive care unit: an observational study. Clin Med Insights Circ Respir Pulm Med. 2019;13: 1179548419871527.
3. Vetrugno L, Bove T. Lung ultrasound estimation of pleural effusion fluid and the importance of patient position. Ann Intensive Care. 2018;8:125.
4. Vetrugno L, Brogi E, Barbariol F. “A message in the bottle.” Anesthesiology. 2018;128(3):677.
5. Griffiths JR, Roberts N. Do junior doctors know where to insert chest drains safetly? Postgrad Med J. 2005;81:456-458.
Dr. Nichole Tanner discusses how to optimize tissue acquisition during EBUS procedures

Nichole Tanner, MD, MSCR, discussed how to optimize tissue acquisition during endobronchial ultrasound (EBUS) procedures at a lecture during American Thoracic Society International Conference. Currently, Dr. Tanner is an assistant professor of medicine in the pulmonary and critical care division at the Medical University of South Carolina and the co-director of the Lung Screening Program at the Hollings Cancer Center in Charleston, South Carolina.
Below is a video and full transcription of her presentation.
5825943360001
brightcove
true
Dr. Tanner is a paid consultant of Cook Medical.
A history of lung cancer diagnosis
MATT REPASY: Okay, welcome to our workshop, “Optimizing tissue acquisition.” I am Matt Repasy. I am a trainer for Cook Medical. Today, you guys are going to hear a short lecture, we’ll take some questions, and then after the lecture we will introduce the workshop stations. You can continue to eat your lunch, but on your chairs here we have some marketing information but, there is also a feedback form, so if you do have an opportunity before you leave the workshop today, fill out the feedback form, and you can just leave it on the chair or hand it to one of the Cook employees as you walk out of the workshop, that would be wonderful. I would like to just do a short introduction of our faculty. We will have two faculty today working the EBUS workshop stations. Dr. Abdul Alraiyes did his residencies in both anesthesia and internal medicine, and did his pulmonary critical care fellowship at Tulane University, and his interventional pulmonary fellowship at Cleveland Clinic. He is currently the assistant professor of medicine and director of interventional pulmonary and critical care medicine at the Cancer Treatment Centers of America north of Chicago. Our speaker for today, Dr. Nichole Tanner, from the Medical School at University of Miami, did her residency and pulmonary critical care fellowship at the Medical University of South Carolina. In 2015, she was awarded the ATS Thoracic Oncology Early Career Achievement Award. She is currently the associate professor medicine and co-director of the Lung Screening Program at the Hollings Cancer Center in Charleston. Without further ado, I will bring Dr. Tanner up to start.
DR. NICHOLE TANNER: Good afternoon. Thank you for having me, and thank you guys for spending lunch here. Hopefully, you will learn a little something while you munch and get some hands-on experience. So, I know everyone wants to get playing with the equipment that we have, so I will try to go through this as quickly as possible. Please feel free to interrupt me with any questions as we go along, okay? So, these are my disclosures. I have taken consulting from Cook and I also have a research grant that the company has funded.
So, let’s start with a case presentation and try to make this a little bit interactive. You have a 54-year-old Hispanic man with a dry cough for about two months. He denies any fevers, chills, or other constitutional symptoms other than a 10-pound weight loss. He is a light smoker with a 10-pack per year smoking history, quit 25 years ago, no significant family history for lung history, and his primary care provider orders a CT scan which looks like this. So, the PET scan demonstrates multiple liver lesions and it was clear that this gentleman had stage 4 disease, and so rather than biopsy the liver, they did EBUS at level 7, and the diagnosis was confirmed of adenocarcinoma. So, the question is what is the next step in managing this patient? Is it A) a palliative care consult with stage 4 disease, B) systemic chemotherapy, or C) send tissue for molecular analysis? C – We’re here at an EBUS workshop, right?
Okay, so I think it’s probably not a surprise to anyone in this room but the treatment for stage 4 non-small lung cancer is changing. Back in the 1980s, there was absolutely no treatment for it, then we started with doublet chemo, and now we’re in the era of targeted therapeutics for stage 4 and immunotherapies, and as you can see with that, survival is improved over the past couple of decades, so it’s really kind of a game-changer for lung cancer.
The importance of actionable targets
Okay, so tissue acquisition in 2017, what’s important? The majority of patients, as we know, at diagnosis with lung cancer before the advent of lung cancer screening, the majority of patients present with locally advanced or distantly advanced disease, so stage 3 and stage 4 is the majority of patients. So, you’ve got your tumor and you need to differentiate whether it’s non-small cell or small cell, and that has treatment implications, obviously. Then you can do some IHC staining to look further because you need to stratify between adenocarcinoma and squamous cell carcinoma because the treatments are different. It can be devastating to give the wrong chemo to a patient with one cancer versus the other.
And now we’re in the era of mutational analysis because we do have actionable targets, and so this statement came out which was a joint guideline from the College of American Pathologists as well as the ISLC, all of this, and what they now say is that non-small cell lung cancer is not acceptable. We need more than that. And so, we do a lot with tissue, right? We get pathology, immunostaining, insight to hybridization, biomarkers, genotypes, and now we’re being asked to get more and more tissue for mutational analysis for all of the drugs that are in the pipeline as well as immune checkpoint inhibitors.
And so, why actionable targets? This is the seminal paper from Dr. Mok that was published in 2009, and as you can see in this one graph here, those with an EGFR mutation positive did really well with targeted therapy, tyrosine kinase inhibitors, and less well with the platinum-based chemotherapy agents. Similarly, those that are negative do better with standard chemo versus the TKI, so this is kind of where it all started.
So here is our case, and then he was treated with erlotinib for an EGFR mutation, and as you can see, a picture is worth a thousand words. So, we’re moving in this era where we can treat very advanced stage 4 disease with targeted therapies if the person is mutated, and if this was my family member, I’m sure all of you would agree you would want to know what the mutations are. So, this is on three months, and you can see things get better. Now I still point out, we still cannot cure lung cancer that’s stage 4, but the length and the duration that people are living is much longer.
Okay, and so what of all these mutations that we are being asked to do? This was an article that was published in 2014 in JAMA by Dr. Chris and colleagues, which was an amalgamation of this Lung Cancer Mutational Consortium, and their goal was to determine the frequency of these oncogenic driver mutations for adenocarcinoma. It was about 14 sites, and you see the day ranges there, and they were trying to collect at least 10 genes. And what they found was that over 1,000 patients were able to be tested for at least one gene, 733 could be tested for all 10, and they found a driver mutation in 64% of this group, and the results used to select therapy in 28% of the time. So, you can see there the survival difference with the orange line in those that received targeted therapies versus those without targeted therapies, and then those who did not have a driver mutation.
An increase in mutations and the creation of new drug therapies
So, the incidence of single driver mutations is going up, I don’t know if you can see that, but we’re looking at the pie and it’s getting split with more and more things. So, as I said before, 64% of the time they found a mutation, which is fantastic as drug companies are out there trying to target these mutations, and hopefully we will have a bigger armamentarium of how we can treat our patients with lung cancer.
I bring this article up because it’s very important, because we did say we can’t cure stage 4 lung cancer, and what if you have someone on a tyrosine kinase inhibitor and then they advance again? So, we do know that those that have the EGFR mutation who are treated with tyrosine kinase inhibitors can develop a resistance mutation, the so-called EGFR T790M mutation, and so this results in tumor resistance to the tyrosine kinase inhibitor. So, this new drug, which is labeled here AZD-9291, was used to test for those people who had this T790M mutation, and as you can see, the response rate to those that were positive to this drug was very good. So, what I’m saying is that if you think about kind of the HIV continuum, and I know we’re a little off from that, but we start with one drug, they develop resistance, and we test again and we find another drug, so we as pulmonologists are going to be asked to continue to test. So, we get that first mutation, EGFR positive, somebody progresses, now we have to again sample that site to test for the next mutation to see if there is another drug. So, you can see how this is going, and we need to find a way to do more with less. So, this study really highlights the importance of repeating biopsy for molecular analysis at the time of disease progression, and any medical oncologist worth his weight will hopefully ask you, or the interventional radiologist depending on the site of recurrence, to obtain another sample.
And so, what about the number of actionable targets? It’s just rising, it’s getting higher and higher and higher, and I haven’t even started to talk about immunotherapy. So, that’s a good transition. I like this little slide, it’s an electron… the microscope. Anyways, it’s really nice. At least once a day, your immune system destroys a cell that would have become cancer if it lived. And so just to kind of review the biology of it, and I think it’s very fascinating, cancer cells have mutations that make them recognizable to the immune system, and the job of our immune system is to clear these rogue cells. However, cancer cells can evade the immune system by expressing certain proteins such as PDL1, and so basically, they cause the immune system to become blind to them and the tumors proliferate. So, if you can inhibit this PDL1 ligand or PD1 interaction, you can restore anti-tumor cell activity, so the T cells become activated and all of a sudden, they can see the cancer cells again, and this can potentially lead to very long-lasting results.
And so nivolumab was the first drug that was approved for this mutation, and you can see there with the arrows where the drug acts. It’s a fully human IG4 Programmed Death immune checkpoint inhibitor. It disrupts this signaling and restores anti-tumor immunity so that your patient’s immune system can effectively go after the tumor. And this has been shown in this New England Journal paper here, and as you can see, this was the inclusion and exclusion criteria, stage 3 or 4 squamous cell cancers, they also did this is adeno, and as you can see they had quite a good enrollment. Primary outcome was overall survival, and the usual secondary outcomes are listed there. And interestingly, they actually tested whether or not the drug worked better depending on the level of expression for PDL1. That’s an immunohistochemistry stain, okay?
And so, what you can see there is that nivolumab had a much better median overall survival versus docetaxel, and the number of deaths was a lot less. So exciting, and I think what the real excitement is is the durability of the response. So, if you look at these plots, even after they are off the drug, they continue to have a response. So, it’s reprogrammed our immune system, the immune system now recognizes the tumor, and it’s a lasting effect if and when they come off the drug. This is just another article that shows the same thing.
Tissue is the issue
So, I just bring this up to go over what’s happening. Stage 1 we still have surgery, stage 2 surgery with chemo, stage 3 radiation with chemo. Depending on what center you’re at, they might offer surgery for stage 3A patients. Stage 4, we now have this huge armamentarium, and in a disease that we thought was universally fatal, we now have targeted therapies and immune checkpoint inhibitors, and it’s really exciting. For once, we can offer our patients something a little bit more.
But really, tissue is the issue. We are taking these very small biopsies with our EBUS needles and we are being asked to do more and more with less and less, and so is the pendulum going to swing from us doing EBUS back to doing a full mediastinoscopy to get a giant chunk of tissue to run all of this? Do we really want to subject somebody with stage 4 disease to an operating room with the risks of anesthesia and postoperative complications all to get tissue? And so really the conundrum because either we do more with less or we get more tissue, and so I think to do more with less, there are a number of assays and new things like next generation sequencing that allow you to run 30 genes with less and less tissue.
And then on the opposite side is our hardware. What do we have in our toolbox to try get to get more tissue? All right, so which of the following provides the best sample for mutational analysis? Is it A) EBUS FNA, B) pleural fluids, or C) CT-guided transthoracic needle aspiration or an ultrasound-guided transthoracic needle aspiration? Anybody? No guesses? All right, EBUS does pretty well, but if you look–oh, I think I have it on the next slide, so we’re going to go through here. So overall, specimen insufficiency rate was low, and they just looked at EGFR mutations here, and I think maybe KRAS, but as I said, we are doing more and more. What we do know is that EBUS has a pretty good sufficiency as it comes to mutational analysis and that, but this prior to the dawn of everything else. Then they went ahead and looked at it for multi-gene mutational analysis using a 22 gage needle, and they were able to do EGFR analysis in 90% of patients, which is great. I think we can do a lot with EBUS samples, and I would refer anybody who is interested to this article in the Journal of Thoracic Oncology in 2011. It gives a nice review of all the different things that you can do with EBUS, from IHC to FISH and PCR, etc.
So, what’s the optimal number passes when you are trying to do something for mutational analysis? How many times do you have to run that needle back and forth? How many samples – A, B, C, D, anyone? C? C, yeah, very good, good answer. So, the number of passes in a station, it varies and it’s really difficult to conclude. It seems to us that three to four samples are adequate, but we don’t know. This study was done by Lonnie Yarmus out of Hopkins in 2013, and they looked at 85 patients diagnosed with adenocarcinoma via EBUS, and they showed that it was about that number of passes. So, adequate with fewer than four passes, adequate with four or more passes, there really was no difference, 96% versus 94%, so really not a huge difference.
The benefit of bi-bevel needles
So, does needle type or size matter as far as this goes? These are three different papers. The first one by Nakagima and colleagues looked at 33 patients and they said there was really no difference in yields, although there seemed to be a better histologic preservation with a 21 gage needle because there was more blood contamination. In this one other trial, they said that the 21 gage is better, and then in the AQuIRE Registry which is the Bronchoscopy Registry, they looked at close to 1,300 patients and they saw that there was no difference in adequacy or yield. But all of this was just for plain diagnosis, all of this was just non-small cell, small cell, adeno versus squamous, so we really don’t have any data about markers in EBUS yet, all of the different things, yeah.
So that brings me to this, which you guys have some information I think there. This is the bi-bevel needle which has two bevels, that’s which it means bi-bevel, and so it kind of acts like a cheese grater. It was first development I think in the GI space for pancreatic lesions, etc., but it kind of goes in and you get some, and then you pull it back and you get some that way. So that thrusting movement of the needle allows you to grate, grate, if you will, some more tissue. And so, this is kind of what it looks like grossly, and I don’t know how many of you in the room do EBUS. Actually, I probably should have asked that. By a show of hands, how many do EBUS? Okay, so a lot of people, and so I don’t know if you all are preparing your own slides or putting the stuff into cytolyt or whatever your pathologist’s choice is, but you see here the ProCore sample just grossly looks bigger. I think you all agree.
And so, this brings me to the trial that I am the lead investigator on, which is a multi-center trial that compares the standard 22 gage needle to the bi-bevel ProCore needle for endobronchial ultrasound. We know the background. As I said, it was used during EUS. It showed that there were larger samples. They used the same technology and developed this needle for the EBUS space. And so, the goal of the study is really to determine which needle is better, and it’s a multi-center randomized control trial of five different sites. We have already enrolled 200 patients with known or suspected lung cancer. Our primary outcome is the number of tumor cells per millimeter squared, and there is a secondary outcome looking at do we have enough tissue, enough tissue to do all this testing, EGFR testing, etc. So, I think that this will answer some questions, and we are soon to report the results. That’s all I can tell you.
So, implications of mutational analysis to the bronchoscopist, I think that it’s really important. When EBUS came around, it kind of revolutionized our involvement in lung cancer staging as pulmonologists, and now we’re being asked to get more and more. I don’t know how many of you have the experiences I do where the oncologist gets on the phone and says, “I need more tissue. I need a core biopsy. I need, I need, I need.” And we’re all working towards, but it’s important really to develop a streamlined and standardized approach to how you get the sample. I think it’s really important to speak to your pathologist locally because everybody does it a little different. I think the expertise of the center that you’re at might dictate what you do, but you want to ensure that the appropriate tests are ordered and that requires communication with the oncologist. Samples of sufficient quality and quantity, you have to kind of keep revisiting, do some quality control, and I think it’s important that we continue to do repeat biopsies when people progress to assess for a TKI resistance, or even clinical trials if you are in a big institution.
So, what’s the turnaround time? It really just depends. For the IHC, the testing is about one to two days, but when you’re talking about next-gen sequencing and all that stuff, if it’s a send-out, it gets expensive and the time gets there, and you have the patient in front of you, or the oncologist does, and they’re trying to treat them, and so working on that is important. But I think around the average is probably 10 to 14 days till you get everything back, depending on what you’re sending it for.
The importance of histologic subtyping and mutational analysis
So, in conclusion, I think histologic subtyping and mutational analysis are critical steps. EBUS FNA is the diagnostic test of choice for lung cancer tissue acquisition for molecular analysis. I think that this novel needle may provide more tissue for marker acquisition, that’s just my gut feeling. I will tell you that I often at these things get questions like, “How do I prepare my sample? What do you do?” There is no standardization, and so the American Thoracic Society and the American College of Chest Physicians as well as the American Society of Clinical Pathologists, or maybe that’s Cytopathologists, they are putting together a statement, a guideline statement actually, to look at that, and I know that it says here in 2015, but if any of you have been involved in guidelines, it takes like, two or three years to come around. So, they’re doing the literature search and going through all that, and I think it will be really helpful to the practicing pulmonologists, but communication is key.
I will say one quick word about the PDL1 markers. I mentioned nivolumab but we also have pembrolizumab in another trial, and I’m not sure why that didn’t make it into my slide deck, I apologize. But there was a trial that was published looking at another immune checkpoint inhibitor, pembrolizumab—say that 10 times fast. What it did show was that those that has a PDL1 expression of greater than 50% on IHC had a much better response to the drug, and so now we are being asked to send tissue for PDL1 expression, and depending on where you practice, that can be a send-out test. The drug companies actually have specific tests developed, like assays for their drug, and then there is a generic version that I think Quest Diagnostics will do, but some of these assays require “core” specimens, and that’s really debatable what a core is. I mean, we call this a coring needle but is this a tissue core, and that’s just a discussion to have with the labs that you are doing these procedures for. But I think we really need to try to optimize how much we can get with EBUS so that we’re not sending these patients back for big surgical procedures to get tissue. And with that, I thank you for your attention. We’ll take some questions, and then afterwards, Matt will tell us about the hands-on course here.
Q&A session
MAN #1: Thanks for a brilliant talk. Just a short question about the lymphomas. Last year, I also asked the same question to you. What’s the place of the ProCore needles in a diagnosis of lymphoma? Thank you.
DR. NICHOLE TANNER: That’s a good question, “What’s the place for the ProCore needles in the diagnosis of lymphoma,” just so that everybody hears that. I had a hard time hearing that. I don’t know the answer to that and I probably didn’t know last year. I think anecdotally, we had some good experience, and maybe my colleague here can comment on that, of getting it, however, there is no trial. I will tell you that one of my colleagues at my institution has put in an investigator-initiated trial to look at just that. It’s a harder thing to test because you don’t really have a lot of lymphoma cases unless you are like in a giant cancer center, and it all becomes tricky, Hodgkin’s on Hodgkin’s, and they need to see the structure, and I think that’s a lot like pathologically driven, etc. At my own institution at the VA, we are constantly having this argument about, “Do you need to extract an entire lymph node to make the diagnosis and do all of that?” And so, I don’t know, and I hope that we get to do that trial because I think it would be really helpful.
MAN #1: There is a meta-analysis that was done for all the studies, and the diagnosis for lymphoma with EBUS can rate from 30% in some studies up to 80%, but still, you are diagnosing lymphoma, but you might not get the specific type, and that’s why you still need the whole lymph node biopsy.
MAN #2: Thank you, very informative talk. I wondered about, you said that the trial results are not yet out for the ProCore needle. Have you guys used the needle for long, and how much difference has it made as far as the histology is concerned? How happy are the pathologists with you when you send the specimens across?
DR. NICHOLE TANNER: That’s a great question. So, we have been using the needle now for almost three years I would say, and my pathologists love it. They do because anecdotally they will just say, “Oh yeah, we got so much here.” Yeah, so we’re in an academic institution and so we’re always enrolling folks in trials, and I will tell you that for some of these chemo trials, they ask for a “core” specimen, and this pass because we say we did with a coring needle, and so I think that has implication for clinical trials and enrollment of patients. So, the pathologists really love it, the oncologists love it because then they can enroll patients in trials, so I like it.
MAN #3: Are the chances of hemorrhage more with this bi-bevel needle?
DR. NICHOLE TANNER: So, the question is, “Are the chances of hemorrhage worse with the bi-bevel needle?” I can tell you we had not had any adverse events with one needle versus the other during the trial. There were no adverse bleeding events in the 200 patients that we enrolled. I guess I could see your point. Theoretically, if you go into the lymph node and it happens to be very vascular, and you poke it once and then you poke it on the way back, you could theoretically have more hemorrhage, but we haven’t had anything horrible happen. I mean, obviously, you probably wouldn’t want to stick that into a vessel so your EBUS skills need to be good to identify the lymph nodes, etc., but we had no bad bleeds. Other questions?
MAN #4: How is ProCore needle compared to the 19 gage needle?
DR. NICHOLE TANNER: I’m sorry, I couldn’t hear that.
MAN #4: So, 22 gage, is that the biggest EBUS needle that you have?
DR. NICHOLE TANNER: Yeah, the ProCore comes in a 25 and a 22.
MAN #4: So, then we also have a 19 gage needle, right?
DR. NICHOLE TANNER: There is not a 19 gage, right, Matt? Okay.
MAN #5: Thank you for the talk. I have read some article talking about the transvascular needle aspiration. Have you tried this kind of needle for transvascular needle aspiration?
DR. NICHOLE TANNER: Have I tried this kind of needle for a vascular?
MAN #5: Yes.
DR. NICHOLE TANNER: Oh, transvascular, no, I have not. I have not. Actually, I think mentally it worries me just a little bit.
MAN #5: So, do you think they are safe?
DR. NICHOLE TANNER: Yeah, I have not tried. I think our group was the first to publish like going through a pulmonary artery to get the lymph node on the other side, I don’t know. I have not tried it, and honestly it makes me nervous.
MAN #5: Yeah.
WOMAN #1: Does this needle change the flexibility of the scope? And is there a learning curve that you expect?
DR. NICHOLE TANNER: Yes, absolutely there is a learning curve, and we will explain that to you at the work stations. It’s not perfect–and no offense, Cook folks–and there are certain stations that make it more difficult to get into. I will tell you the 4L position, unless the lymph node is very large, that’s a technically difficult space to get in with standard EBUS, and so it does change the angle with which the needle exists. It’s a bit more superficial, not as deep as the usual needles that we’re all used, so you have to kind of get used to that. The radial dials are easier to work with. But I will tell you, and I don’t think this is classified, there is a version two of said needle which is much better and in the works. That’s okay I said that? Okay, after I said it. But yeah, I think that there is a lot of hope for the needle.
MAN #6: Recently there have been like mini-forceps that you can go through the needle. How does that compare to this ProCore needle?
DR. NICHOLE TANNER: Yeah, I have not used the mini-forceps. Have you?
DR. ABDUL ALRAIYES: Usually the mini-forceps, it depends on the content of the lymph node. If it is large or necrotic, you might not get as good as aspiration, but the same thing we used it sometimes in patients who are suspicious for lymphoma to see if we get, and we didn’t report much of difference to.
DR. NICHOLE TANNER: Good question. Anyone else? All right, so now for the fun part, Matt.
MATT REPASY: Okay, so we actually have four stations. Two of them will be EBUS stations. They will be using the Fuji scope along with an animal explant model that you will think is real neat. So, they are identical stations, Dr. Tanner at one station, Dr. Alraiyes at another. We also have a station for how to handle the core sample, the larger sample, so sampling techniques, prep techniques, and then this other station is just additional interventional procedures of Cook if you are interested in other Cook procedures. So really, just go ahead and you can start breaking into both stations, all stations, and we’ll open up the workshop.

 Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax. Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax.
Used for the relief of simple, spontaneous, iatrogenic, and tension pneumothorax. Used for emergency relief and temporary management of suspected tension pneumothorax.
Used for emergency relief and temporary management of suspected tension pneumothorax. Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax. Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax. Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax.
Used for the relief of simple, spontaneous, or iatrogenic pneumothorax and tension pneumothorax. Used for the percutaneous introduction of a chest tube for pleural fluid drainage.
Used for the percutaneous introduction of a chest tube for pleural fluid drainage.
 Used to remove fluid from the pericardial sac.
Used to remove fluid from the pericardial sac.
 Used in conjunction with a pneumothorax aspiration catheter if incomplete expansion of the lung persists after mechanical aspiration of air.
Used in conjunction with a pneumothorax aspiration catheter if incomplete expansion of the lung persists after mechanical aspiration of air. Used for connection to other medical devices such as catheters.
Used for connection to other medical devices such as catheters. Used for the connection of an external drainage catheter to a drainage pouch.
Used for the connection of an external drainage catheter to a drainage pouch. Used to provide separate sampling or an infusion port within Thal-Quick chest tubes.
Used to provide separate sampling or an infusion port within Thal-Quick chest tubes.
 He has also submitted letters to the editor regarding the importance of patient position during ultrasound procedures.
He has also submitted letters to the editor regarding the importance of patient position during ultrasound procedures. Dr. Koenig explained that the use of ultrasound prevents a concept commonly known as “clinical and time dissociation.”
Dr. Koenig explained that the use of ultrasound prevents a concept commonly known as “clinical and time dissociation.”
