Dr. Michael Dake et al. recently published a peer-reviewed article analyzing Zilver PTX patient-level data from two large studies with long-term follow-up to determine whether there was an increased mortality risk due to paclitaxel. See the results.
View Article
View Cook Medical’s updated letter to physicians on paclitaxel
During a June 19-20, 2019, meeting of the Circulatory System Devices Panel of the Medical Devices Advisory Committee, the panel addressed concerns about the use of paclitaxel-coated devices to treat PAD. Physicians, industry leaders, professional organizations, and societies presented their findings to the panel.
At that time, the panel agreed that the benefits of paclitaxel-coated devices continued to outweigh the risks and that these devices should remain on the market.
If you have questions or would like more information on our data please visit our Cook Research Incorporated website.
RCT Data Disclosure
In the spirit of full transparency, which is a highly valued principle at Cook Medical, we were the first and only company affected by the FDA’s 2019 letter to release de-identified patient-level data. The only modifications we made to this data were designed to ensure patient privacy and confidentiality are always protected.
-
This Dataset includes additional vital status data for patients who did not originally complete the study. For patients who were found to have died, information on cause of death was not available; therefore, there was also no CEC adjudication or categorization possible for these patients.
-
This Dataset includes additional vital status data for patients who did not originally complete the study; therefore, the panel pack and panel presentation do not reflect this information.
No significant difference between Zilver PTX and PTA/BMS at 5 years.
Cook’s Presentation to FDA Advisory Panel
Presented by: Aaron Lottes, PhD, and Michael Dake, MD | June 2019
The Cook analysis of actual treatment of Zilver PTX patients included the updated status for 92% of patients who were previously lost to follow-up. The all-cause mortality rate for the PTA/BMS group was 15.6% and 18.9% for the Zilver PTX group (p-value = 0.46). The addition of the new data confirmed no mortality signal for Zilver PTX.
View Presentation
Publications
Provided below are links to the selected original Zilver PTX data publications.
Published by: Cardiovasc Intervent Radiol | Dake et al.
“Paclitaxel-coated Zilver PTX drug-eluting stent treatment does not result in increased long-term all-cause mortality compared to uncoated devices.”
View Publication
E-published ahead of Print: J Am Coll Cardiol | Secemsky et al.
“Drug-Eluting Stent Implantation and Long-term Survival following Peripheral Artery Revascularization”
View Abstract
Conference Presentations
Below, we have collected presentations looking at the meta-analysis published in the Journal of the American Heart Association and reviewing our data on Zilver PTX.
LINC 2021 | Presented by: Aloke Finn, MD | January 2021
CIRSE 2019 | Presented by: Michael D. Dake, MD | September 2019
LINC AP 2019 | Presented by: Carlos Mena, MD | March 2019
CRT 2019 | Presented by: Gary Ansel, MD | March 2019
VLF 2019 | Presented by: Michael D. Dake, MD | March 2019
LINC 2019 | Presented by: Michael D. Dake, MD | January 2019
VIVA 2018 | Presented by: Michael D. Dake, MD | November 2018
Essential prescribing information
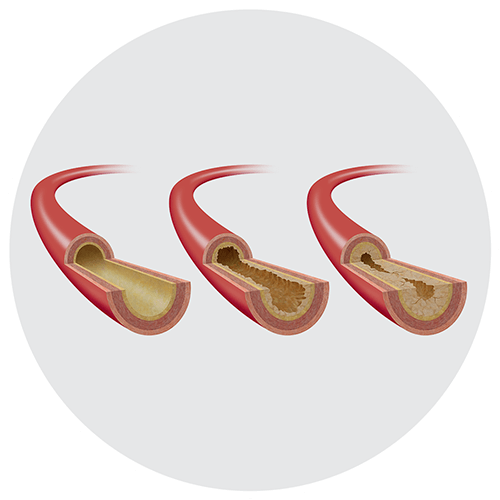
Peripheral arterial disease (PAD) is a serious condition that affects 8-12 million Americans each year.1 PAD occurs when fatty deposits form in arteries that are outside of your heart. These fatty deposits (or plaque) can restrict the flow of blood. PAD often occurs in the legs. In severe cases it can lead to amputation. People who have PAD are at greater risk of suffering a heart attack or stroke.
Cook Medical is committed to helping treat patients who suffer from PAD, allowing patients to get back to living.
What is PAD?
What are the symptoms of PAD?
Most people with PAD don’t show any warning signs. Only one quarter to one third of people who are diagnosed with PAD have any symptoms at all.2 People who do have symptoms often mistake them for signs of aging.
Symptoms include:
- Leg pain when walking
- Numbness or weakness in the legs
- Aching pain in the feet or toes while at rest
- Ulcers or sores in the leg or foot that don’t heal
- Cold legs or feet
- Skin-color changes in the legs or feet
Who is at risk?
PAD can strike anyone, but it is most common in older people. PAD affects up to 20% of people over the age of 70.2 Smoking increases the risk of PAD. Heavy smokers are four times more likely than nonsmokers to develop PAD.2 On average, smokers are diagnosed with PAD 10 years earlier than nonsmokers.2
Risk factors include:
- Diabetes
- Obesity
- High blood pressure
- Lack of exercise
- Family history of hardening of the arteries (atherosclerosis)
- High cholesterol
Treatment of PAD
If you have PAD, your doctor will ask you to exercise more. If you are a smoker, your doctor will ask you to stop smoking. Your doctor may also prescribe drugs to thin your blood and to lower your cholesterol and blood pressure. All of these actions can help to slow the progression of PAD. They can also decrease your chances of having a heart attack or stroke. However, drugs and lifestyle changes alone don’t resolve all cases of PAD. For these patients, angioplasty, stenting, or bypass surgery may be necessary.
Angioplasty
A doctor can use a very small balloon to open up fatty deposits. The doctor uses a needle through the skin to access the artery and then inserts long, thin tools to reach fatty deposits. The doctor threads the deflated balloon through the fatty deposit and then inflates the balloon. After the fatty deposit is opened the doctor deflates and withdraws the balloon.
Stenting
If an artery is still too narrow after angioplasty, or your doctor thinks that the balloon treatment alone won’t help your artery stay open, he may use another tool called a stent. A stent is a small, metal tube that lines the inside of a fatty deposit and keeps the artery propped open. Like balloons, stents are delivered through blood vessels. Once a stent is placed it remains there forever. If an artery closes again, then another procedure may be necessary.
Bypass Surgery
Angioplasty and stenting doesn’t work for all patients. In these patients, doctors may do bypass surgery. Bypass surgery involves cutting open the leg and sewing a new vessel above and below the fatty deposit. The new vessel can be artificial or a vein that is transplanted from another part of the body.
What is Zilver PTX?
Zilver PTX is a drug-coated (drug-eluting) stent. The stent is used to treat the fatty deposits that can restrict blood flow in the largest artery of the thigh. This vessel is called the superficial femoral artery (SFA). The Zilver PTX stent is made of nitinol. Things that are made of nitinol return to their original shape after they are squeezed or bent. Nitinol makes the Zilver PTX stent open up by itself when your doctor puts it in your artery. Stents like Zilver PTX that open up by themselves are called self-expanding stents. Zilver PTX is the first stent with a drug coating that the FDA approved for use outside of the heart.
Benefits of Zilver PTX
Zilver PTX holds open the SFA and delivers the paclitaxel. The drug helps stop tissue growth that could close an artery. If an artery closes again, then another procedure may be necessary. This treatment is highly valued by patients because it improves their quality of life and decreases their need for repeat procedures.
Who should not get a Zilver PTX stent?
The following people should not get Zilver PTX:
- Women who are pregnant, are breast-feeding, or plan to become pregnant in the next five years.
- Patients who cannot take blood thinners (antiplatelet drugs).
- Patients in whom stents cannot be properly placed.
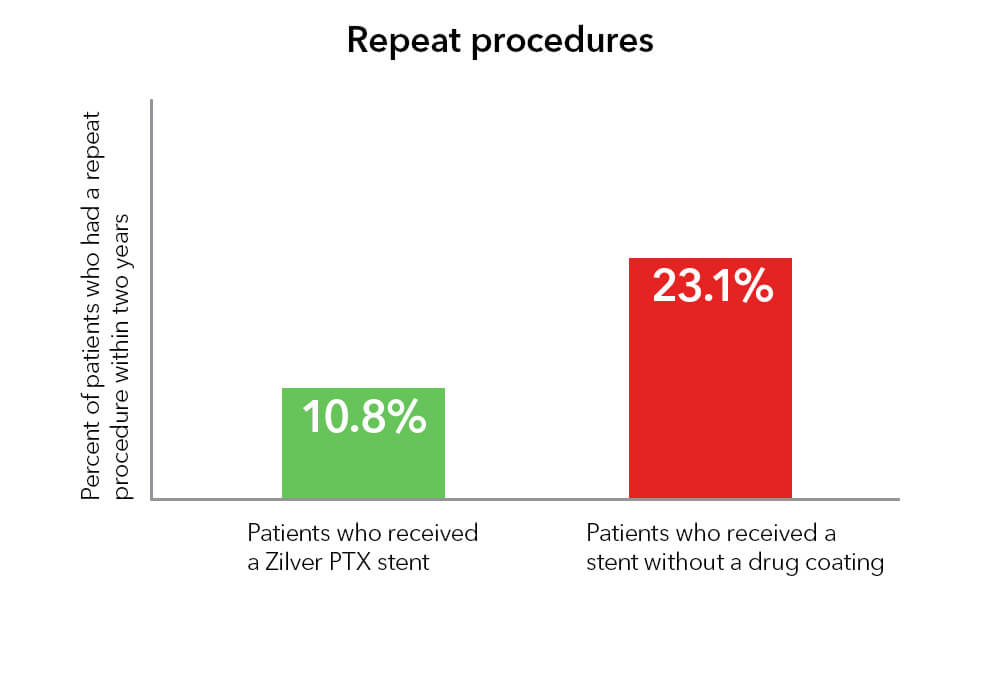
Repeat procedures
Compared with stents that are not coated with a drug, Zilver PTX reduced the number of patients who needed to have a repeat procedure by 53% at two years.3
Two years after treatment, 74.8% of patients who received Zilver PTX and 26.5% of patients who were treated initially with balloons still had open arteries without additional treatment.
4
What is paclitaxel?
Paclitaxel was discovered in 1967 in the bark of the Pacific yew tree. This drug blocks a cell’s ability to divide and is often used to treat cancer. For cancer patients, the paclitaxel dose is large and goes throughout the body. The Zilver PTX stent carries a much smaller paclitaxel dose that is applied directly to the artery wall. Your blood may wash away a small amount of the drug.
How does paclitaxel work?
The paclitaxel on Zilver PTX starts to go into your artery wall as soon as your doctor places the stent. Once inside a cell, paclitaxel blocks parts of cells that are involved in cell division. When the cells can’t divide, the excess growth that can close your artery again is prevented. Over time, a kind of cell growth that is good takes place. The inner lining of the artery grows over the stent and reduces the risk of blood-clot formation.
Risks of paclitaxel
The following could happen to you when you get a drug-coated stent:
- You could have an allergic reaction to paclitaxel.
- You could experience unwanted hair loss.

- Your number of red blood cells could decrease.
- Your bone marrow could produce fewer blood cells.
- You could need a blood transfusion.
- You could experience symptoms in your stomach and intestines.
- Your number of white blood cells or platelets could decrease.
- Your liver function could change.
- The wall of your artery could die or become damaged or inflamed.
- You could experience joint pain or muscle pain.
- Your nerves could become damaged.
After researching paclitaxel for more than 25 years, we have been able to conclude that paclitaxel from the Zilver PTX stent is no longer present in the body after approximately two months.5 Tens of thousands of patients have been treated with the Zilver PTX stent in the SFA for more than 10 years. We have commissioned numerous studies, including one of the world’s largest (479 patients) and longest (5-year follow-up) randomized controlled trials on a device to treat SFA disease.6 We believe such long-term results are critical to understanding how drug-eluting devices treat vascular disease.
- National Institutes of Health. Facts about peripheral arterial disease (P.A.D.). National Heart, Lung and Blood Institute Web site. https://www.nhlbi.nih.gov/health/educational/pad/docs/pad_extfctsht_general_508.pdf. Published August 2006. Accessed April 10, 2019.
- Norgren L, Hiatt WR, Dormandy JA, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). Eur J Vasc Endovasc Surg. 2007;33(suppl 1):S1-S75.
- Ansel G. Zilver PTX randomized trial of paclitaxel-eluting stents for femoropopliteal dis¬ease: 24-month update. Presented at: the Society for Cardiovascular Angiography and Interventions (SCAI) 2011; May 4-7, 2011; Baltimore, Maryland.
- Ask your doctor about the Zilver PTX Instructions for Use (IFU). This document contains full prescribing information, including indications, contraindications, warnings, precautions, and clinical data.
- Dake MD, Van Alstine WG, Zhou Q, et al. Polymer-free paclitaxel-coated Zilver PTX stents – evaluation of pharmacokinetics and comparative safety in porcine arteries. J Vasc Interv Radiol. 2011;22(5):603–610.
- Dake MD, Ansel GM, Jaff MR, et al. Durable clinical effectiveness with paclitaxel-eluting stents in the femoropopliteal artery: 5-year results of the Zilver PTX randomized trial. Circulation. 2016;133(15):1472-1483.
PI-D48307-EN
 Cook Medical recently participated in a collaborative, first-of-its-kind fellows workshop at the Interventional Radiology−Translational Research and Simulation Lab (IR−TRSL) at the Cincinnati Children’s Hospital Medical Center (CCHMC). The innovative workshop was designed for adult and pediatric IR fellows at the University of Cincinnati (UC) Medical Center and CCHMC respectively.
Cook Medical recently participated in a collaborative, first-of-its-kind fellows workshop at the Interventional Radiology−Translational Research and Simulation Lab (IR−TRSL) at the Cincinnati Children’s Hospital Medical Center (CCHMC). The innovative workshop was designed for adult and pediatric IR fellows at the University of Cincinnati (UC) Medical Center and CCHMC respectively.
The fellows program was the brainchild of Dr. Seetharam Chadalavada, Assistant Professor of Radiology, Division of Interventional Radiology at UC Health. To bring his vision to life, Dr. Chadalavada collaborated with Dr. John Racadio, Division Chief of IR at CCHMC, and Nicole Hilvert, Senior Research Assistant and Manager of the IR-TRSL. Together, they devised a course that would utilize the strengths of each facility.
During the half-day course, IR fellows from UC and CCHMC were provided opportunities to experiment with new techniques, fluoroscopy time, and learning about tools and resources … all without practicing on a patient.
“We wanted to develop a course that helps fellows understand the depth and breadth of their toolbox in simulated scenarios that are available in the IR-TRSL,” Dr. Chadalavada said.
According to Hilvert, the lab has historically been used for pre-clinical technology development, device evaluation, physician training, and even occasionally to provide care for animals from the Cincinnati Zoo. “The idea to repurpose the lab to allow more time for fellows training seemed like an ideal inter-hospital partnership,” she said.
To procure the necessary devices for the workshop, Dr. Chadalavada first reached out to Saumya Premachandra, Cook Medical’s manager of the Vista Residents and Fellows Program. He had previously collaborated with Cook on a hands-on program during his fellowship program in 2015 at the Hospital of the University of Pennsylvania.
“It was a natural fit for us to collaborate with UC on this new fellowship program because Cook has such an extensive offering of products,” Saumya said. “The fact that the faculty developed a self-assessment survey to measure improvements in the fellows’ technical and clinical skill set competency made this a unique collaborative experience.”
According to the post-workshop survey, fellows reported that their clinical and technical confidence levels had improved as a result of the course. There was also consensus that they had enjoyed the hands-on experience with new devices and techniques. One of the workshop participants stated that he particularly enjoyed “getting to try things that I haven’t been able to do in fellowship thus far, such as iliac and renal stenting.”
Each said they would recommend that the workshop be incorporated into the UC and CCHMC IR fellowship programs.
“This is the first iteration of something that will be evolving,” Dr. Racadio said. “We will continue to tailor the course to better serve our fellows going forward.”
Dr. Chadalavada said the fellowship experience is vital in the transition to the role of attending.
“One of the most important things to learn in order to become an effective attending physician is how to problem-solve, evaluate, and make decisions,” he said. “The decision-making component is a composite of bookwork, technical skills, resources, experience, technology, and equipment training. All of these culminate into the ability to provide the greatest care for patients.”
For more hands-on opportunities, sign up for one of Cook Medical’s Vista® Education and Training courses.
Dr. Seetharam Chadalavada, Dr. John Racadio, and Nicole Hilvert are not paid consultants for Cook Medical.












 Cook Medical recently participated in a collaborative, first-of-its-kind fellows workshop at the Interventional Radiology−Translational Research and Simulation Lab (IR−TRSL) at the Cincinnati Children’s Hospital Medical Center (CCHMC). The innovative workshop was designed for adult and pediatric IR fellows at the University of Cincinnati (UC) Medical Center and CCHMC respectively.
Cook Medical recently participated in a collaborative, first-of-its-kind fellows workshop at the Interventional Radiology−Translational Research and Simulation Lab (IR−TRSL) at the Cincinnati Children’s Hospital Medical Center (CCHMC). The innovative workshop was designed for adult and pediatric IR fellows at the University of Cincinnati (UC) Medical Center and CCHMC respectively.