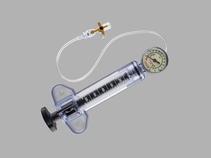
During minimally invasive balloon dilation procedures, the Cook Sphere Inflation Device can be used to inflate the balloon with fluid, to monitor the pressure in the balloon during the procedure, and to deflate the balloon after dilation.
The products on this website are available for sale in the United States. Reference Part Numbers are not the same in all countries/regions. For products available in other countries, please choose one of our other websites from the region selector at the top of the site or contact your local Cook Medical representative.