New agreement expands research for cutting-edge medical technologies, strengthens workforce pipeline

Purdue President Mung Chiang (center, left) and Pete Yonkman, president of Cook Group and Cook Medical, sign a new agreement to advance innovative medical research and technologies that builds upon a 50-year alliance. (Purdue University photo/John Underwood)
WEST LAFAYETTE, Ind. — Purdue University and medical device manufacturer Cook Medical on Thursday (May 7) announced a 5-year, master sponsored research and collaboration agreement (MSRCA) to jointly advance innovative medical technologies, while also significantly expanding the university’s research capabilities and bolstering the biomedical engineering workforce pipeline.
The MSRCA builds upon a legacy of collaborative research excellence between the two institutions dating back more than a half-century.
The agreement creates formal framework for multiple collaborative projects targeting image-guided procedures and advanced medical device manufacturing.
“For half a century, Purdue’s relationship with Cook has advanced biomedicine and life sciences from labs to lives,” said Purdue President Mung Chiang. “This agreement will allow us to significantly accelerate and amplify our collaboration further, creating jobs, talent and life-saving medical innovations right here with another milestone of industry-university partnership.”
The MSRCA establishes a standardized outline for partnering on research, testing and collaborative activities that bridge the gap between laboratory discovery and real-work patient care.
The multifaceted cooperation is designed to streamline and expedite project delivery and make research faster, more efficient and more impactful for both institutions’ research and educational missions.
“Indiana’s life sciences leadership is no accident; it’s the result of intentional choices by visionary people and institutions. Cook is proud to be part of that story. Agreements like this one are how Indiana’s vision becomes reality, keeping world-class talent and investment right here at home,” said Pete Yonkman, president of Cook Group and Cook Medical.
The partnership will engage researchers and resources across the university’s colleges, institutes and departments – connecting academic rigor and research depth to address real-world biomedical demands.
The research agreement advances three of Purdue’s four strategic priorities — Purdue Computes, One Health, and Purdue University in Indianapolis — by bringing together AI and frontier innovation, holistic health research, and expanded opportunities across the West Lafayette and Indianapolis locations and Cook facilities.

Joining in discussion: Jennifer Kerr (BS biomedical engineering ’93, MS basic medical sciences ’95), president of Cook Research Inc., Derek Voskuil, chief operating officer of Cook Medical; and Kevin Otto, the Dane A. Miller Head of the Weldon School of Biomedical Engineering.
“By accelerating the power of transformative computational technologies, the recently forged agreement gives Cook and Purdue the ability to model, simulate and solve biomedical challenges at scales that were previously out of reach,” said Jennifer Kerr, president of Cook Research Inc. and Purdue alum. “By working closely with Purdue, we’re not only pushing the boundaries of technology but also preparing the next generation of biomedical professionals who will change how patients are treated.”
The relationship between Purdue and Cook Medical dates back to collaborations in the 1970s between pioneering Purdue researcher Leslie Geddes, the Showalter Distinguished Professor Emeritus of Biomedical Engineering and one of the founders of Purdue’s biomedical engineering program, and Cook Group founder Bill Cook, whose revolutionary discoveries allowed doctors to conduct minimally invasive procedures.
What began as a shared commitment to advancing medical innovation has grown into a sustained partnership embedded within Purdue Research Park and the Weldon School of Biomedical Engineering.
“The roots of this collaboration run deep, grounded in the visionary work of Leslie Geddes and Bill Cook,” said Arvind Raman, the John A. Edwardson Dean of the College of Engineering. “Together, they built a legacy that transcends generations — one that continues to define the enduring partnership between Purdue University and Cook Medical and its impact on biomedical innovation.”
Following the ceremony, Dean Raman moderated a discussion panel that examined how AI, engineering and medicine are unlocking new capabilities; how biomedical tech is shifting from treatment to prediction; and how these technologies will change everyday health.
Joining in the discussion were Jennifer Kerr (BS biomedical engineering ’93, MS basic medical sciences ’95), president of Cook Research Inc., Derek Voskuil, chief operating officer of Cook Medical; and Kevin Otto, the Dane A. Miller Head of the Weldon School of Biomedical Engineering.
50-year history of biomedical engineering at Purdue
https://engineering.purdue.edu/BME/AboutUs/News/2024/50year-history-of-biomedical-engineering-at-purdue
History of biomedical engineering at Purdue
Video: https://www.youtube.com/watch?v=9ZOJ9wLSbtU
Bringing the latest in medical technologies to the youngest patients
https://www.purdue.edu/newsroom/2023/Q3/bringing-the-latest-in-medical-technologies-to-the-youngest-patients/
About Purdue University
Purdue University is a public research university leading with excellence at scale. Ranked among top 10 public universities in the United States, Purdue discovers, disseminates and deploys knowledge with a quality and at a scale second to none. More than 106,000 students study at Purdue across multiple campuses, locations and modalities, including more than 57,000 at our main campus locations in West Lafayette and Indianapolis. Committed to affordability and accessibility, Purdue’s main campus has frozen tuition 14 years in a row. See how Purdue never stops in the persistent pursuit of the next giant leap — including its integrated, comprehensive Indianapolis urban expansion; the Mitch Daniels School of Business; Purdue Computes; and the One Health initiative — at https://www.purdue.edu/president/strategic-initiatives.
Media contact: Wes Mills, wemills@purdue.edu
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Media contact: Marsha Lovejoy, Marsha.Lovejoy@CookMedical.com

This article is part of our Ask the CMO series, where Cook Medical’s chief medical officer, Dr. John Kaufman, answers questions. Learn more about Dr. Kaufman in his Meet Our Leaders bio.
Q: What are some disease states and interventional therapies that would greatly benefit from iMRI technology?
Dr. Kaufman: Interventional MRI will have the biggest impact in cancer treatment, musculoskeletal disorders, pediatric care, and cardiac interventions.
Cancer is probably one of the biggest areas for contribution for iMRI. Currently MRI is used extensively in the diagnosis, staging and follow-up of patients with cancer, for example liver, prostate, and soft tissue tumors. There are some interventions that are performed on cancer patients under MRI, but because of the technical limitations, it’s really not been pursued on a large scale. Our collaboration with Siemens Healthineers and our development of iMRI-compatible devices could unlock a huge range of cancer therapies for patients that have been previously unavailable.
MRI is a particularly great imaging modality for musculoskeletal diseases because of the ability to differentiate the many soft tissues such as muscles, tendons, cartilage, nerves, and the linings of joints. Biopsies, ablations (thermal destruction of tissue), nerve blocks, and perhaps even spine interventions may be ideal for iMRI.
In keeping with our commitment to find solutions for all patients in need, the potential pediatric applications are very exciting. Interventional MRI could be very impactful for kids who have chronic diseases that that currently require multiple repeat procedures with ionizing radiation. If you can do these procedures in MRI, it may be beneficial in the long term. While we always want to keep x-ray exposure as low as possible, we are less concerned about one or two exposures in adults. But in some children who are getting imaging and image-guided procedures on almost a monthly basis, this can be a big deal.
Additionally, the cardiac world is very interested in iMRI. Cook is not currently deeply involved in the cardiac space, but this may lead us in a new direction. So, these are just a few of the early areas where iMRI could significantly change current procedures. Once we have iMRI tools in everyone’s hands, it will be really exciting to see all the different places it gets applied.
Q: What are some challenges with iMRI?
Dr. Kaufman: Interventional MRI has some technical, procedural, and some logistical challenges.
Technical challenges
The major technical issue is that it is a completely different environment than most interventionalists are used to working in. There is a very strong magnetic field, and everything needs to be compatible with this. You must have dedicated tools that are not attracted to the magnetic field, don’t create a lot of artifact, but that you can still see with MRI. This creates a lot of challenges in terms of creating devices, including some of the standard accessories that you need when you are doing an intervention like monitors and ultrasound machines. All tools need to be specially designed to work in this environment.
Now, that being said, everyone should understand that there is already a a lot of patient support equipment that is designed for more powerful MRI machines than the 0.55T interventional MRI. It is very common, particularly for children, to have MRIs under sedation or general anesthesia. There are already ultrasound machines that work in this environment. It may sound like a daunting challenge to create iMRI-compatible medical devices, but it is really not as great as you might think. Right now, it’s primarily the procedural tools and devices that have led to the limited adoption of iMRI.
Procedural challenges
It’s also difficult procedure-wise to figure out the configuration of working in an MRI. It’s a small space. It has typically been a very confined environment. However, the iMRI machines have a larger bore, and we are working on those tools that will allow them to be easily manipulated in that environment.
Logistical challenges
The logistical challenges are also daunting. There are different safety requirements around MRIs. Patients have to be screened in a different way than they are for other procedures.
Additionally, getting physicians the necessary time on diagnostic magnets for procedures is challenging in most places unless they have a dedicated MRI for intervention. However, the training needed for running the magnets for both intervention and diagnostic components are all things that Cook has planned for and can provide.
How Cook Medical is solving these iMRI challenges
We’re rethinking the way the whole iMRI suite works and solving challenges with iMRI-conditional devices, training modules, installation support, redesigned MRI machines, and more. Pete Polverini, the vice president of Cook’s iMRI division, has viewed these challenges and opportunities in a holistic way. He’s thinking beyond the idea of finding a device that works in a magnet–he’s thinking about this entire environment and what is needed to make this successful. We’re excited to continue working with Siemens Healthineers on creating that environment that overcomes the logistical, procedural, and technical challenges.
Key highlights
- Cook Medical is one of only five suppliers nationally recognized in the inaugural HIRC Member Choice Awards program
- Cook won in the Supplier Transparency category, recognized for its Customer Portal, data dashboards and collaborative Supply Chain Fellowship program
- The award reflects Cook Medical’s ongoing commitment to supply chain resilience and continuity of patient care

Left to right: Travis DeWolfe, Ross Harvey, Emma Strieter, Haley Aud
Bloomington, Ind. — Cook Medical won the Healthcare Industry Resilience Collaborative (HIRC) Member Choice Award at the annual HIRC meeting. Cook won the award in the Supplier Transparency category. This award is an example of Cook’s efforts to improve our customers’ experience.
How winners are chosen
Each year, the HIRC Member Choice Awards honor suppliers nominated by HIRC’s provider members for demonstrable practices that strengthen supply assurance and continuity of patient care. This inaugural program recognized only five suppliers nationally, underscoring the rarity of being nominated and awarded among HIRC’s member network. Awardees were determined by customer nominations and must hold one or more HIRC badges. The nomination process requires customers to rank nominees against five key criteria for the category and to submit five short essay responses — a process that requires thoughtful consideration and time commitment.
Why Cook was chosen for the HIRC Member Choice Award
Cook Medical was nominated for, and won, the Member Choice Award for several reasons, including:
- The Customer Portal, which shares timely, usable data that empowers two-way collaboration and earlier risk detection.
- The Supplier Risk Dashboard, which shows risk disclosures that enable accurate demand planning and forecasting so customers can continue patient care as smoothly as possible
- The Supply Chain Data Management (SCDM) dashboard, which shows data across procurement, production, logistics and distribution networks for more efficient mitigation and purchasing planning
- Cook’s Supply Chain Fellowship program, a Cook-funded fellowship where a recent MBA graduate works with an industry organization to better understand the relationship between suppliers and providers. The research is published to share their learnings with the whole industry
Quotes
“Supply chain resilience starts with visibility, and visibility starts with honest, open communication. We built our supplier portal and our other data dashboards to give our customers the transparent information they need to plan, adapt and keep delivering for their patients. This award is a testament to the teams across Cook who make that possible every day,” said Ross Harvey, vice president of global supply chain at Cook Medical.
“Our Supply Chain Fellowship was a major factor in Cook receiving this award. The bright fellows have already published several insightful reports from their learnings. The fellowship is a chance for Cook and the entire industry to better understand the dynamics between suppliers and providers and how we can refine processes that benefit everyone. And in the spirit of true learning, we share the research freely for everyone to learn from,” said Travis DeWolfe, supply chain solutions manager at Cook Medical.
About HIRC
The Healthcare Industry Resilience Collaborative (HIRC) is a non-profit, member-driven consortium that strengthens healthcare supply-chain resilience to protect continuity of patient care. HIRC brings providers, suppliers, and partners together to turn shared resiliency and transparency standards into sourcing-ready, measurable tools that reduce risk and support real-world procurement and operational decisions.
FAQs
What is the HIRC Member Choice Award?
The HIRC Member Choice Award is an annual recognition program that honors medical device and healthcare suppliers nominated by HIRC’s provider members for practices that strengthen supply assurance and continuity of patient care. This inaugural year, only five suppliers were recognized nationally.
What is Cook Medical’s Customer Portal?
The Cook Medical Customer Portal is our self‑service digital platform that enables customers to view product and order information, monitor availability, initiate returns, and access supply chain insights—helping increase transparency, reduce manual touchpoints, and improve the overall customer experience.
What is Cook Medical’s Supply Chain Fellowship?
The Cook-funded Supply Chain Fellowship pairs a recent MBA graduate with a healthcare provider. The fellow, with no ties to either organization, can perform objective research. Both organizations agree on a specific project in advance of finding a fellow who is best suited with the appropriate skills to problem solve. Fellows are responsible for finding scalable, real-world solutions and sharing those solutions with the industry by creating a white paper. This is a way for their experience to continue to live on and foster innovation within the healthcare supply chain.
How does this award benefit Cook Medical’s customers?
This recognition validates that Cook Medical’s commitment to supply chain transparency is making a real difference for healthcare providers. By sharing actionable data through the Customer Portal, Cook helps customers plan more effectively, mitigate supply risks earlier, and maintain continuity of care for their patients.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
 At Cook Research Incorporated (CRI), being “first” isn’t about recognition—it’s about improving how quickly safe, effective medical devices reach patients.
At Cook Research Incorporated (CRI), being “first” isn’t about recognition—it’s about improving how quickly safe, effective medical devices reach patients.
In 2025, CRI became the first U.S.-based company to earn certification to ISO 14155, an international standard that defines how clinical studies on medical devices should be designed, conducted, and reported. The standard focuses on protecting patients, ensuring high-quality data, and maintaining ethical research practices.
For CRI, pursuing this certification was a natural extension of its role supporting Cook Medical. The global Clinical Affairs team designs and runs clinical studies that help demonstrate whether new medical devices are safe and effective—an essential step before those devices can be approved for patient use.
Why this certification matters
Medical device companies can conduct clinical studies without ISO 14155 certification, as long as they follow applicable regulations. However, certification provides independent verification that those studies meet globally recognized best practices.
“Regulators want to know they can trust the data they’re reviewing,” said Josh Hawkins, who served as CRI’s Quality Assurance director during the certification process. “This certification shows that our processes—from how studies are designed to how data is collected and analyzed—meet a very high standard.”
That confidence can help streamline interactions with regulatory agencies like the U.S. Food and Drug Administration, ultimately supporting more efficient pathways to approval.
What it takes to earn ISO 14155 certification
The certification process is rigorous and comprehensive.
ISO 14155 covers every aspect of a clinical study involving human participants, including:
- How studies are planned and approved
- How research sites and participants are selected
- How data is collected, monitored, and verified
- How results are analyzed and reported
- How patient safety and ethical standards are maintained
To achieve certification, CRI worked with TÜV SÜD, an independent organization that audits companies against international standards.
The effort took about six months of preparation, followed by both remote and in-person audits before certification was awarded. The process included:
- A detailed review of CRI’s procedures and documentation
- A multi-day, on-site audit with independent reviewers
- Interviews with subject matter experts across the organization
- Verification that systems for data collection, storage, and reporting met strict requirements
“It’s truly everything,” said Dan Martin, senior manager of CRI’s Clinical Infrastructure team. “Every step of how a clinical study is run is evaluated—from the first plan to the final report.”
A proactive approach to quality
Previously, CRI demonstrated alignment with ISO 14155 through its broader quality certification (ISO 13485). However, evolving industry rules meant that approach would no longer be sufficient.
Rather than simply removing that reference, CRI chose to pursue standalone certification.
“We wanted to be clear and transparent about the standards we follow,” Hawkins said. “When the opportunity came to be certified directly, we saw it as the right step—not just for compliance, but for leadership.”
Supporting faster access to innovation
While CRI primarily supports internal work for Cook Medical, the impact of this certification extends beyond the organization.
More than just a piece of paper, this certification highlights CRI’s commitment to:
- Consistently design and run studies to the highest quality standard
- Reduce questions from regulators by strengthening the credibility of methods followed and clinical data collected
- Improve efficiency in the approval process
- Support faster access to new medical technologies for patients
A culture of innovation and collaboration
Achieving this milestone required coordination across teams in multiple countries, including contributors from Europe and Australia who participated across time zones.
“It was absolutely an all-hands effort,” Martin said. “We relied on experts across the organization to make this happen.”
The initiative also reflects a broader mindset within CRI and Cook Medical—one focused on continuous improvement and taking on new challenges.
“We don’t just want to follow what’s already been done,” Martin said. “We want to find better ways to get high-quality devices to the patients who need them.”
FAQ
What is ISO 14155?
ISO 14155 is an international standard that outlines best practices for clinical studies of medical devices involving human participants. It ensures studies are conducted ethically, safely, and with reliable data collection and reporting.
Why is this certification important?
Certification provides independent confirmation that a company’s clinical research processes meet global standards. This helps build trust with regulators, healthcare providers, and patients.
Does this change how clinical studies are conducted?
CRI was already following these principles. Certification formally verifies and strengthens those practices through independent review.
How does this benefit patients?
By improving the quality and consistency of clinical research, certification helps ensure that medical devices are evaluated thoroughly and efficiently — supporting faster access to safe and effective treatments.
Who provides the certification?
Independent organizations, sometimes called certification bodies, audit companies against international standards. In this case, CRI worked with TÜV SÜD.
Does this mean devices are approved faster?
Not automatically. All devices must still meet regulatory requirements. However, high-quality, trusted data can help reduce delays during the review process.
Key highlights
- Cook Medical and Siemens Healthineers unveiled the fully integrated Interventional MRI Suite at SIR 2026.
- iMRI enables a minimally invasive interventional practice ideal for precise soft tissue imaging, tumor targeting, biopsy and future ablation therapies, representing the future of radiation-free, image-guided care.
- Attendees at SIR 2026 got to explore the suite, hear from expert interventional radiologists, learn more about the Cook and Siemens Healthineers partnership and listen to panels about the future of iMRI.
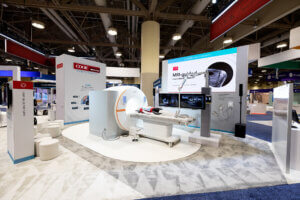
Fully integrated Interventional MRI Suite
Bloomington, Ind. — Cook Medical and Siemens Healthineers unveiled the integrated Interventional MRI (iMRI) Suite at the Society of Interventional Radiology (SIR) 2026 Annual Scientific Meeting in Toronto. The collaboration brings together advanced magnetic resonance (MR) imaging, MR-conditional interventional devices and clinical workflow solutions designed to support minimally invasive procedures in an ionizing radiation-free environment. The unveiling marks a key milestone in the companies’ multi-year effort to advance MRI-guided intervention from concept to clinical reality.
The iMRI Suite provides a comprehensive framework for MRI-guided procedures. The solution combines imaging technology, purpose-built interventional devices, suite planning guidance and clinical education designed to support hospitals adopting MRI-guided interventions. The iMRI ecosystem will use SIR’s VIRTEX, a clinical data registry and analytics platform, to capture data, support continuous learning and help develop evidence-based protocols. The iMRI Suite is a turnkey solution that will give physicians more options for minimally invasive procedures that benefit from superior soft-tissue visualization and a radiation-free environment.
Attendees at SIR 2026 had the opportunity to meet world-renowned clinicians that are advancing clinical care with the newest interventional radiology (IR) techniques. Visitors at the booth got a sneak peek of Cook’s innovative pipeline of products, explored the physical suite environment, participated in demonstration sessions and heard from leading interventional radiologists discussing the emerging role of MRI-guided intervention. The discussions centered on these main topics:
- Procedures that could benefit from real-time MRI guidance, particularly soft-tissue and oncology interventions, such as biopsies and ablations, as well as pediatric and cardiac indications
- Workflow considerations for MRI-guided procedures
- Integration of Cook’s MR-conditional interventional devices with the MAGNETOM Free.XL MRI system from Siemens Healthineers
The iMRI advisory board also discussed the ongoing development of iMRI Centers of Excellence, which are leading academic institutions collaborating with Cook Medical and Siemens Healthineers to advance research, clinical protocols and physician training in MRI-guided intervention. Most recently, Indiana University was named a Center of Excellence in the US. The advisory board will continue planning collaborations with high-profile research facilities across the US and Europe.
Keep up with the latest iMRI developments by following Cook Medical’s iMRI Division page on LinkedIn.
Quotes
“The iMRI Suite is more than a technology; it’s a fundamental shift in how we approach intervention. By bringing together advanced imaging, purpose-built devices, and integrated workflows, we are enabling physicians to see more, treat with greater precision, and ultimately deliver better outcomes without exposing patients or care teams to radiation,” said Peter Polverini III, vice president of Cook Medical’s iMRI divison.
“MRI offers unique advantages for image-guided intervention, particularly for procedures involving soft tissue. Through our collaboration with Siemens Healthineers and leading clinicians, we are working to advance the development of radiation-free interventional approaches,” said Pete Yonkman, president of Cook Medical and Cook Group.
“At Siemens Healthineers, it is our goal to elevate health globally. Magnetom Free.XL is designed to unlock the full potential of MR in the interventional suite, expanding the imaging toolbox by matching the right modality to the right patient at the right time”, said Andreas Schneck, head of Magnetic Resonance at Siemens Healthineers.
About iMRI
MRI provides exceptional soft-tissue visualization and eliminates exposure to ionizing radiation. While MRI has traditionally been used for diagnostic imaging, advances in device design and workflow are enabling its use in minimally invasive procedures. The iMRI Suite was developed to support this transition by creating an integrated environment for MRI-guided interventions.
In September 2025, Cook Medical’s iMRI division and Siemens Healthineers joined forces with leading strategic partners to deliver a fully integrated iMRI Suite. Cook brings procedural, device and training expertise, while Siemens brings world-leading imaging technology. Together, the companies are continuing a multi-year journey aimed at transforming interventional medicine by establishing the iMRI suite as the gold standard for precision, radiation-free procedures.
FAQs
What is Interventional MRI (iMRI)?
Interventional MRI (iMRI) refers to minimally invasive procedures performed using real-time MRI guidance. Unlike traditional interventional imaging modalities such as CT or fluoroscopy, MRI provides superior soft-tissue visualization and does not use ionizing radiation. This enables physicians to target certain tissues more precisely while avoiding radiation exposure for both patients and healthcare providers.
What is the iMRI Suite?
The iMRI Suite is an integrated procedural environment designed to support MRI-guided interventions. The solution combines Siemens Healthineers MRI imaging technology with Cook Medical’s interventional experience and compatible devices, along with suite planning guidance, clinical training and workflow support. The goal is to help hospitals implement MRI-guided procedures safely and efficiently.
What types of procedures could benefit from MRI-guided intervention?
MRI guidance may be particularly valuable for procedures involving soft-tissue structures where visualization is critical. Examples include biopsy and ablation procedures in organs such as the liver, kidney and prostate, as well as certain interventional oncology and pediatric applications. Clinical adoption will continue to evolve as physicians explore new use cases.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
Cook Medical’s Syfonix® Suction Ureteral Access Sheath and the new 7.5 Fr Ascend™ Single-Use Flexible Ureteroscope are now available
Key highlights
- The Syfonix Suction Ureteral Access Sheath is now commercially available in the U.S. and Canada.
- Syfonix allows clinicians to manage suction right at the handle, helping maintain visibility and efficiency during ureteroscopic stone procedures.
- The Ascend Single-Use Flexible Ureteroscope is now available in a new 7.5 Fr size in the United States and Canada.
- Both products are part of Cook Medical’s complete portfolio of disposable urological medical devices. A complete portfolio is an economic advantage for customers on contract with Cook.
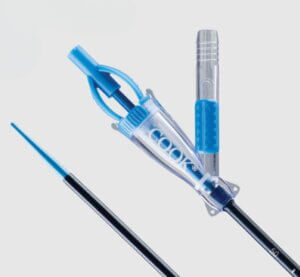
Cook Medical’s Syfonix® Suction Ureteral Access Sheath
Bloomington, Ind. — Cook Medical today announced the commercial launch of two products from its Urology specialty. The Syfonix® Suction Ureteral Access Sheath and the 7.5 Fr Ascend™ Single-Use Flexible Ureteroscope are available in the United States and Canada.
Syfonix is designed to give physicians fingertip control of suction during ureteroscopic kidney stone management procedures. With inner diameter ranges from 9 Fr to 12 Fr, the sheath establishes a working channel to facilitate the passage of endoscopes and ancillary instruments into the urinary tract. It is a single-use device.
Additionally, the Ascend Single-Use Flexible Ureteroscope is now available in a new 7.5 Fr size in addition to the 8.6 Fr size Cook has been offering. As Cook’s pipeline of products includes more products and more sizes, physicians are empowered to treat patients with more precision.
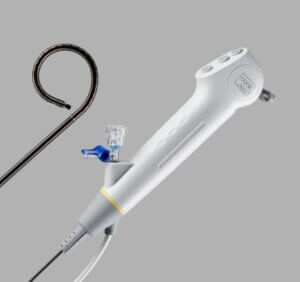
Cook Medical’s Ascend™ Single-Use Flexible Ureteroscope in 7.5Fr
Syfonix and Ascend are compatible products for stone management procedures. Together, these two products are part of Cook Medical’s full portfolio of urological disposables. Cook is proud to be a trusted source for a full line of disposables and the latest laser technology for urological healthcare providers. Having a complete portfolio provides a smoother customer experience for ordering products, especially for customers on contract. To learn more about Cook’s complete stone management portfolio, visit Cook Medical’s Urology specialty page.
Quotes
“Suction sheaths are a critical part of improving stone management procedures. Syfonix gives physicians seamless control of suction without having to break focus or rely on additional staff,” said Johan Lowinger, director of global product management for Urology at Cook Medical. “With the Syfonix sheath now available with the Ascend 7.5 Fr flexible ureteroscope, we’re excited to offer a solution that reflects direct feedback from the urology community.”
“Suction sheaths are one of the newest tools urologists have to help ensure patients get stone-free in a minimally invasive manner. Now that we have added Syfonix to our product portfolio, along with continuing to expand our Vision offering with the addition of the Ascend 7.5 Fr Ureteroscope, we’re giving physicians a full line of products to customize their treatment plan to the specific patient they’re treating. Commercializing these products in the US and Canada is an important milestone for Cook as we work alongside our clinical partners to further advance treatments in stone management,” said Shayna Martin, director of sales for Cook Medical’s Urology specialty.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
Key takeaways:
- What: Cook Medical and Indiana University launch iMRI Center of Excellence to advance radiation-free, MRI-guided medical interventions.
- How: The work spans two complementary initiatives: IU School of Medicine/MIRI medical imaging research infrastructure and the FAMES–Cook engineering initiative for MR-visible devices and to translate iMRI into clinical practice.
- Impact: Creates replicable model for hospitals to implement safer MRI-guided interventions while eliminating radiation exposure for patients and medical staff
Bloomington, Ind. — Cook Medical has selected Indiana University as one of the first Interventional MRI (iMRI) Centers of Excellence. The collaboration is designed to accelerate the advancement, validation, and clinical integration of innovative MRI‑guided technologies and make certain medical procedures safer, more precise, and less invasive for patients.
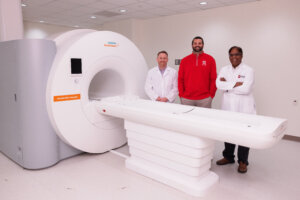
From left to right, Sean Chambers, Director of Research and Development at Cook Medical; David Rosenberg, President and CEO of IU LAB; and Rohan Dharmakumar, Executive Director of the Medical Imaging Research Institute pose for a photo in the iMRI suite. The photo was taken at IU Indianapolis on Wednesday, March 4, 2026. (Photo by Liz Kaye/Indiana University)
The Center of Excellence builds on a five‑year agreement announced in 2025 between Cook Medical and the Indiana University Launch Accelerator for Biosciences (IU LAB) to translate IU research into real‑world health care applications. The collaboration underscores Cook’s broader focus on innovation.
What is the Interventional MRI (iMRI) Center of Excellence?
The iMRI Center of Excellence is a coordinated model that combines device engineering, imaging science, and clinical research infrastructure to speed the development and adoption of MRI‑guided technologies in hospitals and health systems. This practice allows doctors to treat patients without radiation risk while seeing soft tissues in greater detail, enabling more precise and potentially safer interventions.
A blueprint for radiation-free intervention
Indiana University will serve as the model site demonstrating the pathway for translating iMRI into future clinical practice. The university will create a working, real-world environment that hospitals and health systems can tour, learn from, and replicate as MRI-guided care grows in Indiana and beyond. The site will also support physician training and knowledge‑sharing as adoption expands, while helping prepare a new generation of iMRI technologists and clinical staff needed to support MRI-guided procedures nationwide.
“This Center of Excellence allows Indiana University to capitalize on our research strengths and convert them into real change for patients,” said David Rosenberg, president and CEO of IU LAB. “By bringing engineers, imaging experts, and the clinical excellence of the IU School of Medicine together in one place, we can move promising MRI-guided care ideas out of the lab and into hospitals more quickly – all in service of our goal to help patients benefit from safer, radiation-free procedures.”
“Indiana University School of Medicine is committed to making innovations that improve patient care,” said Jay L. Hess, MD, PhD, MHSA, dean of the Indiana University School of Medicine and executive vice president for university clinical affairs at Indiana University. “This partnership with Cook Medical combines world-class imaging, engineering expertise and clinical leadership. Through creating the iMRI Center of Excellence, we can accelerate the development of safer, more effective treatments for patients in Indiana and around the world.”
“We’re excited to name Indiana University as one of the first Centers of Excellence,” said Pete Polverini, vice president, interventional MRI division, Cook Medical. “Our mission for iMRI is bold: to enable safer, more precise, and more effective procedures using interventional MRI, and this collaboration reflects Cook’s accelerated approach to innovation and moving more quickly to bring our new ideas to patients.
Two complementary initiatives: engineering innovation + clinical integration

Ghazal Yoosefian, a Research Assistant at the Medical Imaging Research Institute of the IU School of Medicine, is collaborating with Yanni Pandelidis from Cook Medical in the lab. This photo was taken at IU Indianapolis on Wednesday, March 4, 2026. (Photo by Liz Kaye/Indiana University)
Indiana University and Cook Medical have launched two complementary initiatives spanning the full continuum of iMRI innovation, from advanced engineering of MRI‑visible devices to radiology and imaging research within the IU School of Medicine.
- The IU School of Medicine’s Medical Imaging Research Institute (MIRI) will serve as the clinical and imaging research hub for the Center of Excellence, providing the imaging research and clinical research infrastructure, model iMRI suite, and dedicated personnel needed to validate, integrate, and translate MRI-guided technologies within real‑world procedural processes.
- The FAMES–Cook initiative drives breakthrough engineering innovation by combining materials science, computational modeling, and advanced fiber manufacturing to develop MR‑visible and unique sensing devices designed specifically for interventional MRI environments.
Together, these initiatives connect engineers, imaging experts and physicians into one interdisciplinary system which accelerates translational research while positioning both organizations at the forefront of precision medicine.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
About Indiana University
Indiana University is one of the nation’s leading public research universities, with nearly 90,000 students across seven campuses, two regional academic centers and nine School of Medicine campuses. Since 1820, Indiana University has helped students create brighter futures, while also driving innovation, from breakthroughs in DNA technology to cancer research to trailblazing cultural programs and resources. IU is home to world-class academics with the country’s largest medical school, the world’s first school of philanthropy, the top-ranked Kelley School of Business and O’Neill School of Public and Environmental Affairs, and the Luddy School of Informatics, Computing and Engineering, the nation’s first school of informatics. The university’s campuses are united by IU 2030, an aspirational vision for a bold and ambitious future focused on student success and opportunity, transformative research and creativity, and service to the state of Indiana and beyond. Learn more at iu.edu.
FAQs
Q: What did Cook Medical and Indiana University announce?
A: They established Cook Medical’s first Interventional MRI (iMRI) Center of Excellence at Indiana University to accelerate the development and clinical adoption of MRI‑guided technologies.
Q: What is the purpose of the iMRI Center of Excellence?
A: To build a vertically integrated pipeline spanning engineering, imaging science, and clinical medicine—supporting validation, clinical integration, and translation of MRI‑guided technologies into real workflows.
Q: What are the two initiatives included?
A: The FAMES–Cook engineering initiative and the Interventional MRI Center of Excellence clinical research infrastructure initiative at the IU School of MedicineMedical Imaging Research Institute.
Cook Medical’s latest two Endoscopy products are designed to enhance GI procedures
Key highlights
- Two new Endoscopy products, the Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps, are now available in the U.S. and Canada. The Cold AcuSnare is also available in direct markets in Europe.
- Both products make Cook’s Endoscopy portfolio more robust, especially for GI procedures. Having a more complete portfolio offers additional savings to customers that have contracts for Endoscopy products.
- Cook Medical is proud to offer these innovative products that further simplify product access, especially for customers on contract.
Bloomington, Ind. — Cook Medical has launched two new products within its Endoscopy specialty. The two products, the Cold AcuSnare® Polypectomy Snare and the Caesar® Alligator Rat Tooth Grasping Forceps, are both designed to make GI procedures easier for clinicians. The Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps, are now available in the U.S. and Canada. The Cold AcuSnare is also available in direct markets in Europe.
In addition to the products’ functional benefits, these products make Cook Medical’s Endoscopy portfolio of products more complete. Healthcare organizations ordering these products may be able to get them at a lower cost, as they can order all their endoscopy-related devices from a single vendor.
Quotes
“We like the challenge of balancing a full portfolio with incremental clinical value. The design features of these two new devices, including bi-directional rotatability and hybrid jaws, are tailored to enhance procedural accuracy and ease of use in complex endoscopic interventions.”
–Ben Bailey, director of product management for Cook Medical’s Endoscopy specialty
“By expanding our Endoscopy portfolio, we are empowering clinicians with more features, more treatment options and more economic benefits through comprehensive product offerings. We are excited to share our full pipeline of innovative products.”
–DJ Sirota, senior vice president of Cook Medical’s MedSurg division
About Cold AcuSnare Polypectomy Snare

Cook Medical’s Cold AcuSnare® Polypectomy Snare
For endoscopic resection of diminutive and small polyps in the gastrointestinal tract, use Cook Medical’s Cold AcuSnare product. The Cold AcuSnare Polypectomy Snare—unlike other snares on the market that use diathermic energy to cut endoscopic polyps—is a cold snare, which does not require cauterization to resect the polyps.
There have been recent shifts in clinical guidelines regarding the use of snares. ASGE and ESGE guidelines now recommend cold snaring techniques (without diathermic energy) for all polyps less than 10 mm. Cold snaring shows lower rates of delayed bleeding as compared to hot snaring for polypectomies and mucosal resections.
Among cold snares from other major manufacturers, Cook Medical’s Cold AcuSnare stands out because of its rotatability. The product has 1:1 rotatability so that clinicians can accurately target polyps and perform polyp resection in a precise manner.
About Caesar Alligator Rat Tooth Grasping Forceps

Cook Medical’s Caesar® Alligator Rat Tooth Grasping Forceps
Cook Medical’s new Caesar Alligator Rat Tooth Grasping Forceps are used to remove tissue, stones, stents or foreign bodies during endoscopic procedures and offer the added benefit of two types of jaws: alligator and rat tooth. The jaws’ different shapes allow healthcare professionals to grasp different types and shapes of tissues, stones and foreign bodies successfully. The product also has bi-directional rotatability, which makes it easier to grasp and manipulate tissues, stones and foreign bodies.
The Caesar Alligator Rat Tooth Grasping Forceps can be used for many types of procedures and are an excellent complementary product for repeated GI procedures for removal of pre-placed plastic stent.
FAQs
What is the Cold AcuSnare Polypectomy Snare?
The Cold AcuSnare Polypectomy Snare is intended to be used without diathermic energy, or cauterization, for the endoscopy resection of diminutive and small polyps in the gastrointestinal tract. The snare can be used on polyps less than 10 mm, which are recommended to be removed without cauterization by medical societies (ASGE and ESGE). As part of Cook’s tissue management portfolio, this snare help clinicians identify and focus on the correct tissue to manage and delineate between healthy and diseased tissue.
What are the Caesar Alligator Rat Tooth Grasping Forceps?
These rotating forceps are used in combination with an endoscope to remove tissue, stones, stents or foreign bodies from the digestive tract. The combination of the alligator grasping strength and rat tooth jaw design optimizes grip strength and helps clinicians retrieve even difficult-to-grasp foreign objects. These forceps are also part of Cook’s tissue management portfolio and help clinicians acquire and obtain targeted tissues or stones, stents or foreign bodies.
What does the addition of these products mean for Cook’s Endoscopy portfolio?
Both products make Cook’s Endoscopy portfolio more robust, especially for GI procedures. Having a more complete portfolio is also an economic advantage and offers more savings to customers that have contracts for Endoscopy products.
How can I learn more about using the Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps in my practice?
Contact your local Cook Medical representative to learn more about trying these products in your practice.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.











