FAQ: The laser safety officer (LSO)
In step one of our four-part journey through our procedural product offering, we focused on all things access and our selection of wire guides, needles, and catheters. In this next stage—dilation—we’re highlighting devices that span a breadth of indications—from treating a stricture, to establishing and maintaining a tract, to placing a stent.
Sequential dilation involves inserting a set of dilators that increase in size for a gradual, atraumatic expansion of narrowed or strictured anatomy.
 Used for dilation of male urethral strictures and vesical neck contractures, the S~Curve™ Urethral Dilators are specifically contoured to support the natural curvature of the male urethra. Each dilator has AQ® hydrophilic coating, a lumen that allows precise advancement over a previously positioned wire guide, and a port on the tip that allows for urine drainage to confirm proper placement.
Used for dilation of male urethral strictures and vesical neck contractures, the S~Curve™ Urethral Dilators are specifically contoured to support the natural curvature of the male urethra. Each dilator has AQ® hydrophilic coating, a lumen that allows precise advancement over a previously positioned wire guide, and a port on the tip that allows for urine drainage to confirm proper placement.
 An introducer set facilitates dilation and can be used to introduce a variety of devices depending on the nature of the procedure.
An introducer set facilitates dilation and can be used to introduce a variety of devices depending on the nature of the procedure.
The Desilets-Hoffman Introducer Set is used for retrograde or antegrade ureteral access and subsequent ureteral stent placement. The dilator has a distinct taper on each end: a standard taper for routine access and a pulled (Van Andel) taper for more gradual dilation to facilitate access in obstructed anatomy. The -RB option has a radiopaque band on the delivery sheath to aid in fluoroscopic visualization.
Gaining ureteral access is one thing. Maintaining it is another, and a reinforced access sheath with a removable dilator may be just the device you need as you perform ureteroscopy.
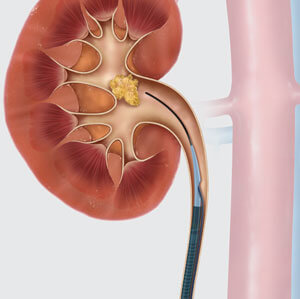 Featuring a tapered dilator and an external hydrophilic coating that creates a low-friction surface, the reinforced Flexor® Ureteral Access Sheath is designed to protect the ureter during repeated instrument exchanges. The Flexor sheath has a PTFE-lined lumen to protect delicate instruments and smaller flexible scopes from damage and is available in a variety of sizes to fit varying scopes.
Featuring a tapered dilator and an external hydrophilic coating that creates a low-friction surface, the reinforced Flexor® Ureteral Access Sheath is designed to protect the ureter during repeated instrument exchanges. The Flexor sheath has a PTFE-lined lumen to protect delicate instruments and smaller flexible scopes from damage and is available in a variety of sizes to fit varying scopes.
Need to perform a nephrostomy? Expand your procedural capabilities with one of our dilation balloons.
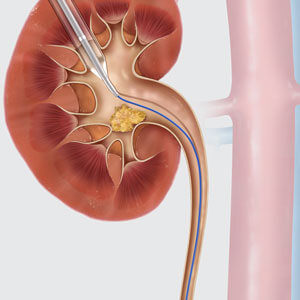 The Ultraxx™ Nephrostomy Balloon Catheter and Set is used to dilate the musculofascia, renal capsule, and parenchyma to establish and maintain a percutaneous nephrostomy tract. The Ultraxx balloon has a rated burst pressure of 20 atm, and the functional dilating portion of the balloon is 15 cm long. The combination of high pressure and balloon length helps you establish a nephrostomy tract in even the most demanding cases. The set is available with either a PTFE or a clear vinyl access sheath. In addition to standard sizes, the Ultraxx is also offered in a smaller 18 Fr option, which allows you to choose the size best suited for the intended procedure.
The Ultraxx™ Nephrostomy Balloon Catheter and Set is used to dilate the musculofascia, renal capsule, and parenchyma to establish and maintain a percutaneous nephrostomy tract. The Ultraxx balloon has a rated burst pressure of 20 atm, and the functional dilating portion of the balloon is 15 cm long. The combination of high pressure and balloon length helps you establish a nephrostomy tract in even the most demanding cases. The set is available with either a PTFE or a clear vinyl access sheath. In addition to standard sizes, the Ultraxx is also offered in a smaller 18 Fr option, which allows you to choose the size best suited for the intended procedure.
Now you’re ready to move on to manipulation and learn about our stone-free solutions that put you in control.
We’re walking you through a procedure from start to finish in a four-part series that takes a closer look at our comprehensive product portfolio. However you choose to get access, Cook has you covered with a diverse selection of wire guides, needles, and catheters.
If you’re performing a ureteroscopy (URS), you may choose to start with a hybrid wire guide and one of our ureteral access catheters. 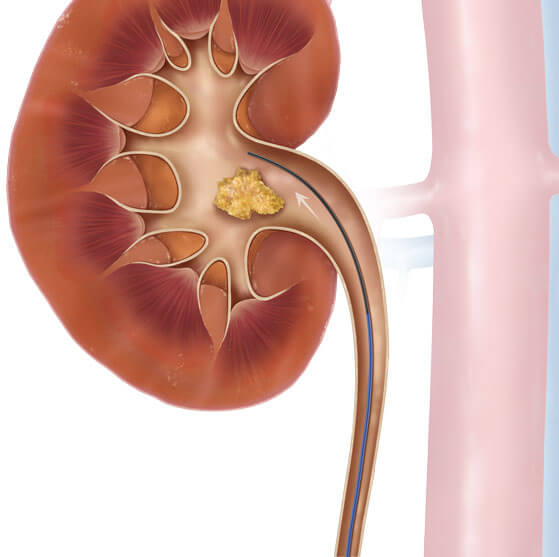
- The Motion™ Hybrid Wire Guide is constructed with a nitinol core and a stainless steel PTFE jacket. Essentially, it’s two wire guides in one. The flexible nitinol distal end has 5 cm of hydrophilic coating and a radiopaque tungsten tip for fluoroscopic visualization. The proximal end of the wire guide is tapered to accommodate scope placement.
Looking for additional options? Explore our selection of nitinol and stainless steel wire guides.
- The Dual Lumen Ureteral Access Catheter may eliminate the need for multiple catheterizations and is used for the injection of contrast medium and anesthetic gel and/or safety wire placement. Available options include a Flexi-Tip® tip configuration, an AQ® hydrophilic coating, and a radiopaque marker.
- The Open-End Flexi-Tip® Ureteral Catheter is used for access and catheterization of the urinary tract to deliver contrast media or irrigation, drain fluids, navigate a tortuous ureter, and advance or exchange a wire guide.
- The Kumpe Access Catheter is composed of a polymer reinforced with a stainless steel braided wire, which provides control, pushability, and torque response. It is used for access and catheterization of the urinary tract, including the delivery of contrast media, the navigation of a tortuous ureter, and the access, advancement, or exchange of wire guides.
Opting for a percutaneous approach?
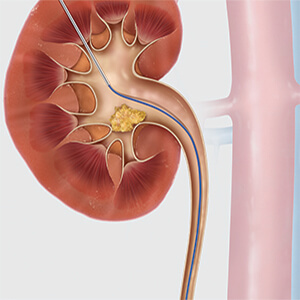
- The Amplatz Fixed Core Wire Guide, constructed of stainless steel, is used to establish a tract and assist in the placement of medical devices. Choose between stiff, extra-stiff, and ultra-stiff options. Tip configurations include flexible and double flexible.
- The Disposable Two-Part Trocar Needle, available in standard and EchoTip® options, is used for percutaneous access. The EchoTip needle is designed to provide enhanced visualization of the needle tip under ultrasound.
- The Amplatz Needle Holder with Silicone Inserts is used to direct standard Cook disposable needles during needle puncture access procedures. It prevents the stylet from backing out of the cannula during puncture and also keeps your hands from radiation exposure during the puncture procedure.
- The Renal Access Cobra Catheter is used percutaneously to direct a 0.038 inch (0.97 mm) diameter wire guide to a specific renal area for the delivery of contrast media through the catheter and for the access, advancement, or exchange of wire guides. The catheter features Torcon® catheter material with an inner stainless steel braid.
Jump ahead in the procedure and check out our selection of manipulation devices.
Talk to your Cook Medical representative to learn more about these products.
In 1998, Cook Medical released the very first extractor made with nitinol,1 an innovative alloy that retains its preformed shape. We called that device the NCircle® Nitinol Tipless Stone Extractor. It was also the first truly tipless basket design. That’s two game-changing innovations in one device.
As we expanded our nitinol offering to include an occlusive umbrella, a multiwire net-like design, and a device that serves as both a basket and a grasper, we addressed some of the more challenging aspects of stone extraction, shaped the baskets accordingly, and continued the trend of alluding to our basket’s unique capabilities in our product names. We literally say what we mean, starting with the uppercase N that speaks to the innovative technology of nitinol.
See our selection of “N-ovations” for yourself.
 Encircle (v.) – to surround
Encircle (v.) – to surround
Because it is truly tipless, the NCircle® Nitinol Tipless Stone Extractor can be positioned flush with the mucosa to surround a stone. Note: Also available in Delta Wire® and helical designs.
 Encompass (v.) – to enclose
Encompass (v.) – to enclose
The multiwire design of the NCompass® Nitinol Stone Extractor produces a netting effect that can help you enclose smaller fragments and prevent stone migration.
 Engage (v.) – to interact directly
Engage (v.) – to interact directly
The NGage® Nitinol Stone Extractor is the original, hybrid basket. Its grasper-like design allows you to directly target a stone and secure it for removal or reposition and release it to be fragmented.
 Entrap (v.) – to catch
Entrap (v.) – to catch
The NTrap® Stone Entrapment and Extraction Device features a bowl-like basket that is designed to catch fragments and minimize stone migration during lithotripsy.
 Enforce (v.) – to obtain with force
Enforce (v.) – to obtain with force
The NForce® Nitinol Helical Stone Extractor is constructed of Delta Wire to help you obtain stones with increased radial force and greater radial dilation.
One of the perks of our nitinol offering is that it gives a physician options to proceed with confidence, no matter the stone. We’ve repurposed a few designs for our percutaneous (Perc) baskets. In 2002, we introduced the Perc NCircle® Nitinol Tipless Stone Extractor. Modeled after the NCircle, and with a stainless-steel cannula and ergonomic handle, the Perc NCircle is specifically designed for percutaneous nephrolithotomy (PCNL). We’ve since added two more Perc extractors to give you additional options for renal calculi.
Experience the shape-memory benefits of nitinol and choose from one of our 26 extractor configurations.
- Monga M. Ureteroscopy: Indications, instrumentation & technique. New York: Humana Press; 2013:180.
 Whether your facility rents or owns a laser system (or multiple systems), you must designate a laser safety officer (LSO) to monitor and enforce your facility’s laser safety program for all laser systems, not just holmium and thulium, and across all specialties, not just urology.
Whether your facility rents or owns a laser system (or multiple systems), you must designate a laser safety officer (LSO) to monitor and enforce your facility’s laser safety program for all laser systems, not just holmium and thulium, and across all specialties, not just urology.
We’ve put together the following frequently asked questions to ensure you take the necessary steps to maintain a safe working environment in your facility.
Who is the laser safety officer (LSO)?
The LSO is an individual, often with a clinical background, who is already serving in another capacity at your facility. They have the authority and responsibility to evaluate, monitor, and enforce the control of laser hazards, and may suspend, restrict, or terminate the operation of a laser system. Their additional responsibilities are listed below.
What are the LSO’s responsibilities?
Responsibilities include, but are not limited to, the following:
- Establish and maintain a safety program in accordance with industry standards
- Classify all lasers under jurisdiction
- Evaluate work area hazards
- Implement control measures
- Recommend protective equipment
- Review warning signs and equipment labels
- Oversee adequate safety education and training
- Perform medical exams as required
- Ensure that necessary records are maintained
- Perform regular audits, surveys, and inspections of safety features and control measures
- Develop a plan for accident response, medical assistance, and documentation
- Approve laser system operations
Who determines the industry standards?
Laser safety standards are regulated by the following agencies:
The ANSI Z136.3 Standard—“Safe Use of Lasers in Health Care”—provides “guidance for the safe use of lasers for diagnostic, cosmetic, preventative, and therapeutic applications where bodily structure or function is altered or symptoms are relieved,” and details the responsibilities of the LSO.
If renting a laser system, does the company providing the laser also provide the LSO?
No. Your facility will be required to designate an on-site LSO to assess compliance of third-party vendor lasers and equipment, maintenance records, staff credentials, and performance in accordance with facility policies for treatment areas.
Does the LSO have to run the laser at all times?
No. The role of an LSO is to support laser safety education and training for all healthcare personnel working with or around laser systems. An employee who isn’t LSO-certified may operate the laser system under the direction and supervision of the LSO.
How should a health system with multiple locations be managed?
The LSO may appoint a deputy laser safety officer (DLSO) to perform the duties of the LSO. For a health system with multiple facilities in various locations, multiple DLSOs may be assigned. A DLSO can be a laser user, laser operator, or someone else who has been trained and is familiar with your laser safety program. The LSO may also designate a laser safety site contact (LSSC) or laser safety specialist (LSS) to oversee safe laser use across multiple locations within a health system. These roles are explained in more detail in the ANSI Standard.
What LSO training is available?
Comprehensive training courses are available from the following providers:
Information about laser safety standards was gathered from the resources listed above. Cook Medical is not affiliated with any of the regulating organizations or training companies mentioned in this post, and the links provided are not promotional endorsements. Please consult with these agencies for the most up-to-date information regarding laser safety standards.
 Used for dilation of male urethral strictures and vesical neck contractures, the S~Curve™ Urethral Dilators are specifically contoured to support the natural curvature of the male urethra. Each dilator has AQ® hydrophilic coating, a lumen that allows precise advancement over a previously positioned wire guide, and a port on the tip that allows for urine drainage to confirm proper placement.
Used for dilation of male urethral strictures and vesical neck contractures, the S~Curve™ Urethral Dilators are specifically contoured to support the natural curvature of the male urethra. Each dilator has AQ® hydrophilic coating, a lumen that allows precise advancement over a previously positioned wire guide, and a port on the tip that allows for urine drainage to confirm proper placement. An introducer set facilitates dilation and can be used to introduce a variety of devices depending on the nature of the procedure.
An introducer set facilitates dilation and can be used to introduce a variety of devices depending on the nature of the procedure. Featuring a tapered dilator and an external hydrophilic coating that creates a low-friction surface, the reinforced Flexor® Ureteral Access Sheath is designed to protect the ureter during repeated instrument exchanges. The Flexor sheath has a PTFE-lined lumen to protect delicate instruments and smaller flexible scopes from damage and is available in a variety of sizes to fit varying scopes.
Featuring a tapered dilator and an external hydrophilic coating that creates a low-friction surface, the reinforced Flexor® Ureteral Access Sheath is designed to protect the ureter during repeated instrument exchanges. The Flexor sheath has a PTFE-lined lumen to protect delicate instruments and smaller flexible scopes from damage and is available in a variety of sizes to fit varying scopes. The Ultraxx™ Nephrostomy Balloon Catheter and Set is used to dilate the musculofascia, renal capsule, and parenchyma to establish and maintain a percutaneous nephrostomy tract. The Ultraxx balloon has a rated burst pressure of 20 atm, and the functional dilating portion of the balloon is 15 cm long. The combination of high pressure and balloon length helps you establish a nephrostomy tract in even the most demanding cases. The set is available with either a PTFE or a clear vinyl access sheath. In addition to standard sizes, the Ultraxx is also offered in a smaller 18 Fr option, which allows you to choose the size best suited for the intended procedure.
The Ultraxx™ Nephrostomy Balloon Catheter and Set is used to dilate the musculofascia, renal capsule, and parenchyma to establish and maintain a percutaneous nephrostomy tract. The Ultraxx balloon has a rated burst pressure of 20 atm, and the functional dilating portion of the balloon is 15 cm long. The combination of high pressure and balloon length helps you establish a nephrostomy tract in even the most demanding cases. The set is available with either a PTFE or a clear vinyl access sheath. In addition to standard sizes, the Ultraxx is also offered in a smaller 18 Fr option, which allows you to choose the size best suited for the intended procedure.













