Cook Medical’s Syfonix® Suction Ureteral Access Sheath and the new 7.5 Fr Ascend™ Single-Use Flexible Ureteroscope are now available
Key highlights
- The Syfonix Suction Ureteral Access Sheath is now commercially available in the U.S. and Canada.
- Syfonix allows clinicians to manage suction right at the handle, helping maintain visibility and efficiency during ureteroscopic stone procedures.
- The Ascend Single-Use Flexible Ureteroscope is now available in a new 7.5 Fr size in the United States and Canada.
- Both products are part of Cook Medical’s complete portfolio of disposable urological medical devices. A complete portfolio is an economic advantage for customers on contract with Cook.
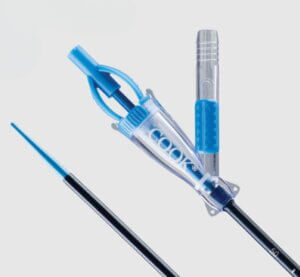
Cook Medical’s Syfonix® Suction Ureteral Access Sheath
Bloomington, Ind. — Cook Medical today announced the commercial launch of two products from its Urology specialty. The Syfonix® Suction Ureteral Access Sheath and the 7.5 Fr Ascend™ Single-Use Flexible Ureteroscope are available in the United States and Canada.
Syfonix is designed to give physicians fingertip control of suction during ureteroscopic kidney stone management procedures. With inner diameter ranges from 9 Fr to 12 Fr, the sheath establishes a working channel to facilitate the passage of endoscopes and ancillary instruments into the urinary tract. It is a single-use device.
Additionally, the Ascend Single-Use Flexible Ureteroscope is now available in a new 7.5 Fr size in addition to the 8.6 Fr size Cook has been offering. As Cook’s pipeline of products includes more products and more sizes, physicians are empowered to treat patients with more precision.
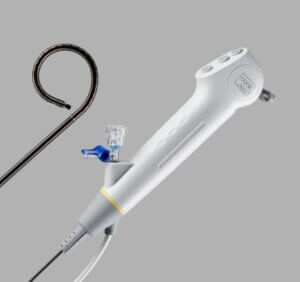
Cook Medical’s Ascend™ Single-Use Flexible Ureteroscope in 7.5Fr
Syfonix and Ascend are compatible products for stone management procedures. Together, these two products are part of Cook Medical’s full portfolio of urological disposables. Cook is proud to be a trusted source for a full line of disposables and the latest laser technology for urological healthcare providers. Having a complete portfolio provides a smoother customer experience for ordering products, especially for customers on contract. To learn more about Cook’s complete stone management portfolio, visit Cook Medical’s Urology specialty page.
Quotes
“Suction sheaths are a critical part of improving stone management procedures. Syfonix gives physicians seamless control of suction without having to break focus or rely on additional staff,” said Johan Lowinger, director of global product management for Urology at Cook Medical. “With the Syfonix sheath now available with the Ascend 7.5 Fr flexible ureteroscope, we’re excited to offer a solution that reflects direct feedback from the urology community.”
“Suction sheaths are one of the newest tools urologists have to help ensure patients get stone-free in a minimally invasive manner. Now that we have added Syfonix to our product portfolio, along with continuing to expand our Vision offering with the addition of the Ascend 7.5 Fr Ureteroscope, we’re giving physicians a full line of products to customize their treatment plan to the specific patient they’re treating. Commercializing these products in the US and Canada is an important milestone for Cook as we work alongside our clinical partners to further advance treatments in stone management,” said Shayna Martin, director of sales for Cook Medical’s Urology specialty.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
Cook Medical’s latest two Endoscopy products are designed to enhance GI procedures
Key highlights
- Two new Endoscopy products, the Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps, are now available in the U.S. and Canada. The Cold AcuSnare is also available in direct markets in Europe.
- Both products make Cook’s Endoscopy portfolio more robust, especially for GI procedures. Having a more complete portfolio offers additional savings to customers that have contracts for Endoscopy products.
- Cook Medical is proud to offer these innovative products that further simplify product access, especially for customers on contract.
Bloomington, Ind. — Cook Medical has launched two new products within its Endoscopy specialty. The two products, the Cold AcuSnare® Polypectomy Snare and the Caesar® Alligator Rat Tooth Grasping Forceps, are both designed to make GI procedures easier for clinicians. The Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps, are now available in the U.S. and Canada. The Cold AcuSnare is also available in direct markets in Europe.
In addition to the products’ functional benefits, these products make Cook Medical’s Endoscopy portfolio of products more complete. Healthcare organizations ordering these products may be able to get them at a lower cost, as they can order all their endoscopy-related devices from a single vendor.
Quotes
“We like the challenge of balancing a full portfolio with incremental clinical value. The design features of these two new devices, including bi-directional rotatability and hybrid jaws, are tailored to enhance procedural accuracy and ease of use in complex endoscopic interventions.”
–Ben Bailey, director of product management for Cook Medical’s Endoscopy specialty
“By expanding our Endoscopy portfolio, we are empowering clinicians with more features, more treatment options and more economic benefits through comprehensive product offerings. We are excited to share our full pipeline of innovative products.”
–DJ Sirota, senior vice president of Cook Medical’s MedSurg division
About Cold AcuSnare Polypectomy Snare

Cook Medical’s Cold AcuSnare® Polypectomy Snare
For endoscopic resection of diminutive and small polyps in the gastrointestinal tract, use Cook Medical’s Cold AcuSnare product. The Cold AcuSnare Polypectomy Snare—unlike other snares on the market that use diathermic energy to cut endoscopic polyps—is a cold snare, which does not require cauterization to resect the polyps.
There have been recent shifts in clinical guidelines regarding the use of snares. ASGE and ESGE guidelines now recommend cold snaring techniques (without diathermic energy) for all polyps less than 10 mm. Cold snaring shows lower rates of delayed bleeding as compared to hot snaring for polypectomies and mucosal resections.
Among cold snares from other major manufacturers, Cook Medical’s Cold AcuSnare stands out because of its rotatability. The product has 1:1 rotatability so that clinicians can accurately target polyps and perform polyp resection in a precise manner.
About Caesar Alligator Rat Tooth Grasping Forceps

Cook Medical’s Caesar® Alligator Rat Tooth Grasping Forceps
Cook Medical’s new Caesar Alligator Rat Tooth Grasping Forceps are used to remove tissue, stones, stents or foreign bodies during endoscopic procedures and offer the added benefit of two types of jaws: alligator and rat tooth. The jaws’ different shapes allow healthcare professionals to grasp different types and shapes of tissues, stones and foreign bodies successfully. The product also has bi-directional rotatability, which makes it easier to grasp and manipulate tissues, stones and foreign bodies.
The Caesar Alligator Rat Tooth Grasping Forceps can be used for many types of procedures and are an excellent complementary product for repeated GI procedures for removal of pre-placed plastic stent.
FAQs
What is the Cold AcuSnare Polypectomy Snare?
The Cold AcuSnare Polypectomy Snare is intended to be used without diathermic energy, or cauterization, for the endoscopy resection of diminutive and small polyps in the gastrointestinal tract. The snare can be used on polyps less than 10 mm, which are recommended to be removed without cauterization by medical societies (ASGE and ESGE). As part of Cook’s tissue management portfolio, this snare help clinicians identify and focus on the correct tissue to manage and delineate between healthy and diseased tissue.
What are the Caesar Alligator Rat Tooth Grasping Forceps?
These rotating forceps are used in combination with an endoscope to remove tissue, stones, stents or foreign bodies from the digestive tract. The combination of the alligator grasping strength and rat tooth jaw design optimizes grip strength and helps clinicians retrieve even difficult-to-grasp foreign objects. These forceps are also part of Cook’s tissue management portfolio and help clinicians acquire and obtain targeted tissues or stones, stents or foreign bodies.
What does the addition of these products mean for Cook’s Endoscopy portfolio?
Both products make Cook’s Endoscopy portfolio more robust, especially for GI procedures. Having a more complete portfolio is also an economic advantage and offers more savings to customers that have contracts for Endoscopy products.
How can I learn more about using the Cold AcuSnare Polypectomy Snare and the Caesar Alligator Rat Tooth Grasping Forceps in my practice?
Contact your local Cook Medical representative to learn more about trying these products in your practice.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.

This article is part of our Ask the CMO series, where Cook Medical’s chief medical officer, Dr. John Kaufman, answers questions. Learn more about Dr. Kaufman in his Meet Our Leaders bio.
Q: You have worked with many other medical device companies and many people in the industry, such as sales reps, trainers, product managers, and regulatory scientists. From a physician’s perspective, how did Cook differentiate itself?
Dr. Kaufman: The reason that I was so excited to join Cook is that for my entire career I always felt that anyone that I worked with from Cook is more of a partner with aligned interests toward doing the best thing for the patient than a transactional relationship. I’ve never felt with Cook that there was a pressure to use something that somebody needed to fill a quota for or convince me to switch to something. It was never that way. It is always with Cook that I felt aligned, to the extent that I could trust anyone from Cook to say, “We don’t have the thing you are looking for, but this other company does.” When I first then began investigating or meeting people as potentially becoming part of Cook that culture was very clear.
What is different about Cook is that, although that culture may be present at other companies, it is such a strong presence throughout all of Cook. There is a shared alignment of goals between those of us who are taking care of people and trying to do the best thing for the person in front of us regardless of other factors. Overall, that is how Cook operates.
Obviously, we are a company, we have to somehow make enough money to stay afloat and make enough money to take care of the people who work for us as well. So, decisions can’t always be purely altruistic. We do have to make some of those decisions, but that is always in the framework of is this really the right thing to do, not just for us but for the patients of physicians.
What is an area that could potentially be improved on?
What could we do better? I think everyone has a different answer based on their own perspective, and as Cook customer it would be great if we could get new or updated devices through the pipeline and into clinical practice as fast as possible. We have so much fantastic stuff in the wings and I want it now! On a serious note, we also need to focus on preserving our unique culture and relationships with physicians as we navigate the complex and highly regulated world that we now operate in. These two things have been drivers for Cook from the beginning and are in a sense timeless.

This article is part of our Ask the CMO series, where Cook Medical’s chief medical officer, Dr. John Kaufman, answers questions. Learn more about Dr. Kaufman in his Meet Our Leaders bio.
Q: How do you balance your connection with Cook with what is the best option or device for your patient?
Dr. Kaufman: I do love working for Cook. It is one of the best things that has ever happened to me professionally, if not the best thing. And I also love taking care of patients and I so appreciate the ability to do both things at the same time. It is not a thing I take for granted at all, but it is an incredible privilege.
The culture at Cook and the culture of medicine are very similar: You do what is best for the patient in front of you.
Don’t do what is best for you or what might be best for your friend or the company you work for or the hospital you are working for or the professional organization that you are a member of. You do what is right for the person in front of you and that guides you all the time.
That prevents really any issue from arising of, “Should I be using a Cook catheter or a non-Cook catheter?” Cook catheters are the best catheters — they just are. So, I don’t have any qualms about using what I think is the best device. There are other devices that other people do better than us and I will use them because I think it is the right thing. I’m glad that it is evident that I am excited to work for Cook as it is an awesome group of people and an awesome organization.

L-R: Rick Simms (Cook Medical, National Manager, GPO Account Executives), Amber Pastorek (Cook Medical, Account Executive Manager), Glenn Coleman (Premier, Chief Financial Officer & Chief Administrative Officer), Bob Stanley (Cook Medical, National GPO Account Executive), Brian Majoy (Cook Manager, Account Executive Manager), Chad Wissner (Cook Medial, Account Executive), Bruce Radcliff (Premier, President of Supply Chain Services) and Michael Alkire (Premier, President and CEO)
Bloomington, Ind. — Cook Medical is honored to receive a 2025 Legacy Supplier Award from Premier, Inc. This supplier award recognizes Cook’s deep connections and commitments to hospitals, supply chain leaders and other healthcare organizations.
Premier is a leading healthcare improvement and technology company that unites an alliance of more than 4,350 U.S. hospitals and health systems. Each year, Premier announces award winners at their Breakthroughs Conference. The awards in various categories go to organizations that demonstrate unwavering commitment to delivering top value products and services designed to continually improve patient care.
Cook Medical was honored to be among a select number of organizations that received the 2025 Legacy Supplier Award. Companies earn this award for long-standing support of Premier members through exceptional local customer service and engagement, value creation through clinical excellence and commitment to lower costs. Supplier Legacy Award winners have a tenure of more than three years as a Premier contracted supplier.
What separates Cook from other suppliers is our Customer Portal, which makes the ordering experience more convenient and efficient for customers. Cook also offers a supplier risk dashboard and a biweekly global supply chain report.
“For more than two decades, Cook Medical has been proud to collaborate with Premier,” said Bob Stanley, a national GPO account executive with Cook’s BusinessCare Integration team.”We are truly honored to receive this award from Premier. It reflects our shared mission and our dedication to delivering value to Premier’s membership. We’re excited about what lies ahead and continuing to grow with our customer’s evolving needs.”
To learn more about the advantages of partnering with Cook Medical, visit our BusinessCare Integration site.
About Premier, Inc.
Premier is a leading technology-driven healthcare improvement company, providing solutions to two-thirds of all healthcare providers in the U.S. Playing a critical role in the rapidly evolving healthcare industry, Premier unites providers, suppliers, payers and policymakers to make healthcare better with national scale, smarter with actionable intelligence and faster with novel technologies. With integrated data and analytics, collaboratives, supply chain solutions, consulting and other services, Premier enables better care and outcomes at a lower cost.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.

Some Cook Urology products that are included in the contract with Vizient
Bloomington, Ind. — Cook Medical has been awarded a contract from Vizient for our portfolio of urology devices. This agreement provides increased access to Cook’s urology products for Vizient clients while further strengthening Cook’s decades-long, physician-focused connections with hospitals across the country. Vizient is the nation’s largest provider-driven healthcare performance improvement company.
Through the contract, Vizient clients will experience streamlined ordering and delivery process and continue to receive excellent clinical and supply chain support from Cook Medical representatives. This agreement also allows physicians to continue treating patients with Cook products they know and trust at competitive price points and reliable product availability.
The urology products in this contract include:
“This contract is another milestone in our collaborative relationship,” said Rob Faulkner, senior director of global sales for Cook Medical’s MedSurg division. “We are excited to continue working with Vizient and making our comprehensive line of urological disposables more accessible to physicians and patients.”
In addition to this contract, Cook has previously earned recognition from Vizient. In 2023, Cook earned Vizient Supply Assurance Supplier of the Year Award. To learn more about the advantages of partnering with Cook Medical, visit our BusinessCare Integration site.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
CAUTION – INVESTIGATIONAL DEVICE. Limited by United States law to investigational use.
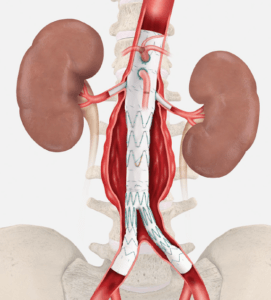
Cook Medical’s investigational ZFEN+ device
Bloomington, Ind. — Cook Medical has enrolled the final patient in the global clinical study of the ZENITH® FENESTRATED+ Endovascular Graft (ZFEN+). This milestone signifies the completion of patient recruitment in the pivotal study and demonstrates Cook’s commitment to innovating medical devices for the treatment of aortic diseases.
The investigational ZFEN+ clinical study is being conducted under an Investigational Device Exemption (IDE) approved by the U.S. Food and Drug Administration (FDA) and with authorization for a clinical investigation under Medicines and Healthcare products Regulatory Agency (MHRA). This clinical study will assess the safety and effectiveness of the ZFEN+ used in combination with the investigational Zenith® Universal Distal Body 2.0 Graft (Unibody2), the investigational Bentley BeGraft Balloon-Expandable FEVAR Bridging Stent Graft System (BeGraft) and the commercially available Zenith® Spiral-Z® AAA Iliac Leg Graft (ZSLE).
“Now that enrollment is complete, we are one step closer to understanding the outcomes of patients treated with ZFEN+ and, ultimately, to advancing care for patients with complex abdominal and thoracoabdominal aneurysms,” said Johnny LeBlanc, director, product management, Aortic at Cook Medical. “Reaching this milestone is a direct reflection of the dedication shown by our investigator partners, study coordinators, and—most importantly—the patients who supported the study with their time and trust. We look forward to analyzing the data and sharing the results with the clinical community.”
The ZFEN+ is predicated on the commercially available Zenith Fenestrated (ZFEN) AAA Endovascular Graft but extends the proximal margin of aneurysmal disease that can be treated endovascularly to include patients with more complex aortic disease involving one or more of the major visceral arteries. The ZFEN+ is an endovascular graft which includes up to 5 precisely located fenestrations, or a combination not to exceed a total of 5 made up of fenestrations and 1 scallop (cut-outs from the proximal margin of the endograft material) to accommodate visceral vessels. Physicians can order fenestrations and scallops specifically to match the patient’s unique anatomy. Overall, the ZFEN+ allows for the endovascular treatment of patients with aortic aneurysms and maximizes the seal zone to exclude the aneurysm. The product was granted Breakthrough Designation from the FDA in 2021.
“Reaching full enrollment in this important clinical study is a major milestone for Bentley and a significant step toward making our BeGraft Bridging stent available to patients in the United States. It underscores the strength of our collaboration with Cook Medical and reflects our shared commitment to advancing innovative vascular solutions that improve patient outcomes,” said Sebastian Büchert, Bentley CEO.
“Amazing work from the clinical research team on the enrollment of the final ZFEN+ patient. The clock is now ticking for us to have a patient specific solution for complex aortic aneurysms that will optimize seal without compromise,” said Dr. Gustavo Oderich, the global principal investigator of the clinical study.
Additional information about the clinical study will be available at www.clinicaltrials.gov.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
CAUTION: Investigational Device. Limited by United States law to investigational use.
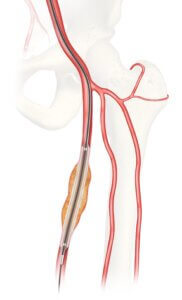
Cook Medical’s investigational Advance Evero™ 18 Everolimus-Coated PTA Balloon Catheter (Evero DCB)
Bloomington, Ind. — The U.S. Food and Drug Administration (FDA) has granted approval for Cook Medical to initiate an Investigational Device Exemption (IDE) study on the Advance Evero™ 18 Everolimus-Coated PTA Balloon Catheter (Evero DCB).
The clinical study will assess the safety and effectiveness of Evero DCB when compared to commercially available paclitaxel-coated balloons for the treatment of peripheral artery disease (PAD). Currently there are no everolimus-coated balloon devices commercially available in the US, and this IDE study is the first U.S. head-to-head evaluation of everolimus- and paclitaxel-coated balloons to treat lesions of the superficial femoral and popliteal arteries.
The EVERO Trial is a prospective, U.S. multi-center, stratified, blinded, randomized control trial with a parallel pharmacokinetic (PK) study. Cook intends to enroll 410 patients in the pivotal trial and 30 patients in the PK evaluation. The primary safety endpoint is a composite of freedom from device- or procedure-related death at 30 days, freedom from target limb major amputation at 12 months, and from target lesion revascularization (TLR) at 12 months. The primary effectiveness endpoint is primary patency, defined as peak systolic velocity ratio (PSVR) ≤2.4 at 12 months and freedom from clinically driven TLR (CD-TLR) at 12 months.
“Advance Evero 18 leverages our deep understanding and history of innovation in drug elution therapies for peripheral arterial disease,” said Dr. John Kaufman, Cook Medical’s chief medical officer. “The head-to-head study design will provide the answers that physicians and patients want. I’m confident that Advance Evero will be an excellent complement to our dedicated PAD portfolio aimed at improving long-term clinical outcomes for these patients.”
Additional information about the clinical study will be available at www.clinicaltrials.gov. You can also learn more about Cook’s portfolio of peripheral intervention products.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
News coverage of this announcement
Endovascular Today: Cook’s Advance Evero 18 Everolimus-Coated Balloon IDE Study Plan Approved by FDA
Vascular News: FDA grants approval for IDE study on Advance Evero 18 everolimus-coated balloon catheter









