Key takeaways:
- What: Cook Medical and Indiana University launch iMRI Center of Excellence to advance radiation-free, MRI-guided medical interventions.
- How: The work spans two complementary initiatives: IU School of Medicine/MIRI medical imaging research infrastructure and the FAMES–Cook engineering initiative for MR-visible devices and to translate iMRI into clinical practice.
- Impact: Creates replicable model for hospitals to implement safer MRI-guided interventions while eliminating radiation exposure for patients and medical staff
Bloomington, Ind. — Cook Medical has selected Indiana University as one of the first Interventional MRI (iMRI) Centers of Excellence. The collaboration is designed to accelerate the advancement, validation, and clinical integration of innovative MRI‑guided technologies and make certain medical procedures safer, more precise, and less invasive for patients.
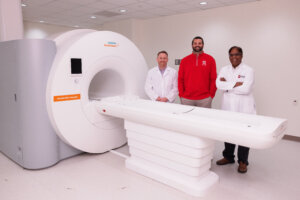
From left to right, Sean Chambers, Director of Research and Development at Cook Medical; David Rosenberg, President and CEO of IU LAB; and Rohan Dharmakumar, Executive Director of the Medical Imaging Research Institute pose for a photo in the iMRI suite. The photo was taken at IU Indianapolis on Wednesday, March 4, 2026. (Photo by Liz Kaye/Indiana University)
The Center of Excellence builds on a five‑year agreement announced in 2025 between Cook Medical and the Indiana University Launch Accelerator for Biosciences (IU LAB) to translate IU research into real‑world health care applications. The collaboration underscores Cook’s broader focus on innovation.
What is the Interventional MRI (iMRI) Center of Excellence?
The iMRI Center of Excellence is a coordinated model that combines device engineering, imaging science, and clinical research infrastructure to speed the development and adoption of MRI‑guided technologies in hospitals and health systems. This practice allows doctors to treat patients without radiation risk while seeing soft tissues in greater detail, enabling more precise and potentially safer interventions.
A blueprint for radiation-free intervention
Indiana University will serve as the model site demonstrating the pathway for translating iMRI into future clinical practice. The university will create a working, real-world environment that hospitals and health systems can tour, learn from, and replicate as MRI-guided care grows in Indiana and beyond. The site will also support physician training and knowledge‑sharing as adoption expands, while helping prepare a new generation of iMRI technologists and clinical staff needed to support MRI-guided procedures nationwide.
“This Center of Excellence allows Indiana University to capitalize on our research strengths and convert them into real change for patients,” said David Rosenberg, president and CEO of IU LAB. “By bringing engineers, imaging experts, and the clinical excellence of the IU School of Medicine together in one place, we can move promising MRI-guided care ideas out of the lab and into hospitals more quickly – all in service of our goal to help patients benefit from safer, radiation-free procedures.”
“Indiana University School of Medicine is committed to making innovations that improve patient care,” said Jay L. Hess, MD, PhD, MHSA, dean of the Indiana University School of Medicine and executive vice president for university clinical affairs at Indiana University. “This partnership with Cook Medical combines world-class imaging, engineering expertise and clinical leadership. Through creating the iMRI Center of Excellence, we can accelerate the development of safer, more effective treatments for patients in Indiana and around the world.”
“We’re excited to name Indiana University as one of the first Centers of Excellence,” said Pete Polverini, vice president, interventional MRI division, Cook Medical. “Our mission for iMRI is bold: to enable safer, more precise, and more effective procedures using interventional MRI, and this collaboration reflects Cook’s accelerated approach to innovation and moving more quickly to bring our new ideas to patients.
Two complementary initiatives: engineering innovation + clinical integration

Ghazal Yoosefian, a Research Assistant at the Medical Imaging Research Institute of the IU School of Medicine, is collaborating with Yanni Pandelidis from Cook Medical in the lab. This photo was taken at IU Indianapolis on Wednesday, March 4, 2026. (Photo by Liz Kaye/Indiana University)
Indiana University and Cook Medical have launched two complementary initiatives spanning the full continuum of iMRI innovation, from advanced engineering of MRI‑visible devices to radiology and imaging research within the IU School of Medicine.
- The IU School of Medicine’s Medical Imaging Research Institute (MIRI) will serve as the clinical and imaging research hub for the Center of Excellence, providing the imaging research and clinical research infrastructure, model iMRI suite, and dedicated personnel needed to validate, integrate, and translate MRI-guided technologies within real‑world procedural processes.
- The FAMES–Cook initiative drives breakthrough engineering innovation by combining materials science, computational modeling, and advanced fiber manufacturing to develop MR‑visible and unique sensing devices designed specifically for interventional MRI environments.
Together, these initiatives connect engineers, imaging experts and physicians into one interdisciplinary system which accelerates translational research while positioning both organizations at the forefront of precision medicine.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
About Indiana University
Indiana University is one of the nation’s leading public research universities, with nearly 90,000 students across seven campuses, two regional academic centers and nine School of Medicine campuses. Since 1820, Indiana University has helped students create brighter futures, while also driving innovation, from breakthroughs in DNA technology to cancer research to trailblazing cultural programs and resources. IU is home to world-class academics with the country’s largest medical school, the world’s first school of philanthropy, the top-ranked Kelley School of Business and O’Neill School of Public and Environmental Affairs, and the Luddy School of Informatics, Computing and Engineering, the nation’s first school of informatics. The university’s campuses are united by IU 2030, an aspirational vision for a bold and ambitious future focused on student success and opportunity, transformative research and creativity, and service to the state of Indiana and beyond. Learn more at iu.edu.
FAQs
Q: What did Cook Medical and Indiana University announce?
A: They established Cook Medical’s first Interventional MRI (iMRI) Center of Excellence at Indiana University to accelerate the development and clinical adoption of MRI‑guided technologies.
Q: What is the purpose of the iMRI Center of Excellence?
A: To build a vertically integrated pipeline spanning engineering, imaging science, and clinical medicine—supporting validation, clinical integration, and translation of MRI‑guided technologies into real workflows.
Q: What are the two initiatives included?
A: The FAMES–Cook engineering initiative and the Interventional MRI Center of Excellence clinical research infrastructure initiative at the IU School of MedicineMedical Imaging Research Institute.
 Cook Medical and Siemens Healthineers today announced a strategic commercial partnership aimed at setting a new standard for interventional medicine. The collaboration combines the power of Siemens Healthineers real-time magnetic resonance imaging with Cook Medical’s deep interventional procedure expertise, along with new medical devices developed by Cook specifically for the MRI environment. Together, the companies are creating the Interventional MRI (iMRI) Suite1, the first integrated, ionizing radiation-free solution intended to transform how clinicians diagnose, plan and perform minimally invasive procedures.
Cook Medical and Siemens Healthineers today announced a strategic commercial partnership aimed at setting a new standard for interventional medicine. The collaboration combines the power of Siemens Healthineers real-time magnetic resonance imaging with Cook Medical’s deep interventional procedure expertise, along with new medical devices developed by Cook specifically for the MRI environment. Together, the companies are creating the Interventional MRI (iMRI) Suite1, the first integrated, ionizing radiation-free solution intended to transform how clinicians diagnose, plan and perform minimally invasive procedures.
“MRI in the interventional suite has always been a concept with tremendous clinical potential, but it has also faced significant barriers. At Siemens Healthineers, we are committed to breaking those barriers to unlock new clinical opportunities and advance patient care. Together with Cook Medical, we’re leading the way into a new era where iMRI becomes a precise radiation-free standard of care,” said Andreas Schneck, head of Magnetic Resonance at Siemens Healthineers.
By leveraging MRI’s unparalleled soft tissue contrast, iMRI enables precise guidance for minimally invasive interventions, particularly in areas such as interventional oncology for soft tissue tumors, pediatrics, and structural heart procedures, where accuracy and safety are paramount. This approach allows clinicians to navigate complex procedures with confidence while eliminating exposure to ionizing radiation for both patients and healthcare teams.
This first-of-its-kind collaboration delivers a turn-key solution designed to accelerate iMRI adoption and advance clinical impact in interventional radiology. It encompasses suite planning, an MRI scanner designed for interventions1, MRI-specific medical devices, specialized training, and ongoing clinical support. Cook Medical brings extensive procedural knowledge, innovative device design, and comprehensive educational programs, while Siemens Healthineers contributes its world-class MR imaging technologies and services, including dedicated interventional planning software.
The integrated iMRI suite reflects a shared commitment to innovation and collaboration, offering clinicians a complete framework, from conceptual suite design to staff training, to enable the adoption of this transformative technology for enhanced patient care.
“Our partnership with Siemens Healthineers is about more than device integration; it’s about revolutionizing interventional procedures,” said Peter Polverini, vice president of Cook Medical’s iMRI Division. “By merging the top-tier MRI systems of Siemens Healthineers with Cook’s advanced devices and expertise, we’re unlocking high-precision treatments that elevate patient care and empower clinicians.”
1 The product is currently under development and not available for sale in the U.S.A. Its future availability cannot be guaranteed.
Disclaimer: The product names and/or brands referred to are the property of their respective trademark holders.
For more information about Cook Medical’s iMRI division, visit CookMedical.com/divisions/iMRI and follow the journey on iMRI LinkedIn.
Further information on the iMRI suite can be found here.
Media contacts
Cook Medical
Marsha Lovejoy
+1 812 320 6903; marsha.lovejoy@cookmedical.com
Siemens Healthineers
Stefanie Haug
+49 173 635 8240; stefanie.haug@siemens-healthineers.com
Visit the Siemens Healthineers Press Center.
Subscribe to our “Medtech matters” newsletter on LinkedIn.
 Bloomington, Ind. — Gayle Karch Cook, historical preservation activist and cofounder of the Cook companies alongside husband Bill Cook, passed away at 91 this morning. She is survived by her son Carl and daughter-in-law Marcy and their granddaughter, Eleanor.
Bloomington, Ind. — Gayle Karch Cook, historical preservation activist and cofounder of the Cook companies alongside husband Bill Cook, passed away at 91 this morning. She is survived by her son Carl and daughter-in-law Marcy and their granddaughter, Eleanor.
“Gayle’s spirit of innovation was evident in everything she did. Whatever she was involved in, she left it better than she found it. This was true of her philosophy for the ever-growing company, her extensive efforts in renovating historically significant places, and her daily interactions with other people. That’s her inspiring legacy that we will keep striving for,” said Pete Yonkman, president of Cook Medical and Cook Group.
In 1963, Bill and Gayle Cook created a small company named Cook Incorporated that manufactured three simple medical devices used to treat patients in less invasive ways than common surgical techniques of the time. Gayle was the company’s first quality control employee, meticulously checking the devices Bill made after he finished work each night.
That small company grew into what is today Cook Group, a family of ethical and entrepreneurial companies. Cook Group includes the flagship company, Cook Medical, a global company that invents, manufactures, and delivers devices for treating conditions in almost every system of the body. Cook Group also includes companies in the life sciences, property management, resort, and service businesses.

Bill, Carl and Gayle Cook
The family planned ahead for this event, and nothing will change for employees moving forward. Carl Cook, Gayle and Bill’s son, has been CEO of Cook Group since his father’s passing in 2011. Company leadership will remain the same with Pete Yonkman as president of Cook Group and Cook Medical and Steve Ferguson as Chairman of the Board.
Gayle Karch Cook, born March 1, 1934, in Evansville, Indiana, made excellence her goal in every aspect of her life. She was salutatorian of Bosse High School when she graduated in 1952, and she earned a Bachelor of Fine Arts from Indiana University in 1956. After graduating, she began her career in advertising in Chicago. After marrying her husband, Bill, they moved to Bloomington, Indiana. There, in the spare bedroom of their apartment, they officially began the company that would later become Cook Medical. The company’s focus has always been on the patient, with a goal to make surgeries minimally invasive.
 She was committed to creating a vibrant community, as she also served on the board of directors for the Star of Indiana Drum and Bugle Corps, the Bloomington Antique Mall, the Nature Conservancy, Indiana Chapter, and the board of trustees of the Indiana University Foundation.
She was committed to creating a vibrant community, as she also served on the board of directors for the Star of Indiana Drum and Bugle Corps, the Bloomington Antique Mall, the Nature Conservancy, Indiana Chapter, and the board of trustees of the Indiana University Foundation.
Gayle was an avid supporter of historic preservation, and she was heavily involved in many restoration projects. She spearheaded about 70 historic preservation and economic revitalization projects, including the much-lauded West Baden Springs and French Lick resorts; Indianapolis’ Central Avenue Methodist Church; and Beck’s Mill and Cedar Farm complex in southern Indiana. The first property she restored and refurnished was the Colonel William Jones House, built in 1835. The house, which is on the National Register of Historic Places, is now a museum open to the public as a State Historic Site. Gayle restored historic sites to their original state, including the James Cochran house, the Illinois Central Freight Depot, the 8-story Graham Hotel, and more than 17 structures in the central square of Bloomington, Indiana.
 In addition to her involvement in the restoration work itself, Gayle served on the boards of many restoration organizations. She was on the advisory committee of the Wylie House Museum, the Board of Governors of the Monroe County Historical Museum, the board of the Historic Landmarks Foundation of Indiana, and was an appointee of the State Historic Preservation Review Board. She was also a founder of the Monroe County History Center Annual Garage Sale which has become a staple in the Bloomington community. With every project, Gayle’s philosophy was to restore not just the site itself, but also the character and beauty of the location for present and future generations. Historic preservation wasn’t just good business; it was an investment in the community.
In addition to her involvement in the restoration work itself, Gayle served on the boards of many restoration organizations. She was on the advisory committee of the Wylie House Museum, the Board of Governors of the Monroe County Historical Museum, the board of the Historic Landmarks Foundation of Indiana, and was an appointee of the State Historic Preservation Review Board. She was also a founder of the Monroe County History Center Annual Garage Sale which has become a staple in the Bloomington community. With every project, Gayle’s philosophy was to restore not just the site itself, but also the character and beauty of the location for present and future generations. Historic preservation wasn’t just good business; it was an investment in the community.
Gayle authored and published two books detailing her passion for Indiana’s history. The first, A Guide to Southern Indiana, was published in 1972. The other, Monroe County in Focus: Portrait of an Indiana County, was published in 1990.
A Phi Beta Kappa graduate of IU, Gayle earned a Bachelor of Arts degree in fine arts in 1956. She and her late husband were generous, far-sighted benefactors of IU’s Jacobs School of Music, School of Education, School of Medicine and Kelley School of Business, as well as supporting IU Athletics, the Wylie House Museum, the Wells Scholars program and research in the College of Arts and Sciences.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
Leveraging Cook’s history of innovative medical devices in a new procedural setting
Bloomington, Ind. — Cook Medical today announced the launch of its new Interventional Magnetic
Resonance Imaging (iMRI) division, marking a significant step forward in its commitment to transforming how complex procedures and unmet clinical needs are addressed.
Unlike conventional tools repurposed for MRI use, Cook Medical is developing a portfolio of minimally invasive devices that have been purposefully engineered, over years of research and refinement, to meet the unique demands of interventional MRI environments. This approach is especially critical in pediatrics, oncology, and pediatric structural heart interventions, where precision and safety are of paramount importance. The new division marks a significant step toward transforming how clinicians diagnose, intervene, and care for patients.
The iMRI division collaborates closely with physicians, hospitals, and strategic partners to seamlessly integrate real-time magnetic resonance imaging. This empowers clinicians to visualize anatomy and instruments simultaneously, without the risks associated with ionizing radiation.
“We know that real progress happens when we listen to the people who use our products every day. The iMRI division is another example of Cook’s commitment to working alongside physicians to solve real challenges and improve lives,” said Pete Yonkman, president of Cook Medical and Cook Group. “We’re proud to support clinicians with technology that helps them do their best work for patients while advancing patient care.”
Rooted in a philosophy of prioritizing patients and physicians, Cook Medical’s iMRI division reflects the company’s longstanding commitment to meaningful, patient-centered innovation. Partnering with premier companies creating state-of-the-art imaging technology, the division aims to empower clinicians with the tools they need for improved outcomes.
Cook Medical appointed Peter Polverini as the new Vice President of the Interventional MRI division. Pete brings over 20 years of experience in advancing medical technology and fostering strong healthcare partnerships.
“I’m proud to lead a team building on years of collaboration with physicians to uncover real clinical needs. This division was born from those insights, where our devices can partner with better visualization and precision guidance to improve patient outcomes. Our mission is bold: to enable safer, more innovative, and more effective procedures through the use of interventional MRI. This is a paradigm shift in medicine, and we’re just getting started,” Polverini said.
About Cook Medical
At Cook Medical, we are passionate about making unique, quality medical devices and connecting with people to improve lives. Founded on inventing, manufacturing, and delivering medical devices, we provide healthcare professionals with the tools they need to help their patients return to living.
Our commitment to innovation involves bringing new products to market and keeping existing products relevant to a changing healthcare landscape. We believe in using our business to help people and communities thrive by creating inclusive, supportive, and healthy environments.
We are proud of our history of innovative firsts and the impact we have on patients and communities. With headquarters in Bloomington, Indiana, and manufacturing facilities and offices in various global locations, we challenge ourselves to maintain a global perspective while focusing on local impact.
Follow Cook Medical at CookMedical.com and on LinkedIn.
For more information about Cook Medical’s iMRI division, visit CookMedical.com/divisions/iMRI and follow the journey on iMRI LinkedIn.
Media Contact:
Marsha Lovejoy
Senior Manager, Global Public Relations
Cook Medical
812.320.6903
Marsha.Lovejoy@CookMedical.com
Please join us as we highlight some of our leaders, learn about their experiences, and hear how they are talking about the positive changes we’re making to meet our vision of the future at Cook. Follow along for the full Meet Our Leaders series.
Meet Mark Breedlove, Senior Vice President, Vascular Division
Please join us as we highlight some of our leaders, learn about their experiences, and hear how they are talking about the positive changes we’re making to meet our vision of the future at Cook. Follow along for the full Meet Our Leaders series.
Meet Ross Harvey, Vice President of Global Supply Chain
Please join us as we highlight some of our leaders, learn about their experiences, and hear how they are talking about the positive changes we’re making to meet our vision of the future at Cook. Follow along for the full Meet Our Leaders series.
Meet Christa Curtis, Vice President Global Marketing and Communications.
 Bloomington, Ind. — Furthering their commitment to relentlessly delivering new products and services to address unmet customer needs, today Cook Medical announced a strategic investment in the urology space. Zenflow is a medical device company developing a minimally invasive treatment for urinary obstruction caused by enlarged prostate, or benign prostatic hyperplasia (BPH).
Bloomington, Ind. — Furthering their commitment to relentlessly delivering new products and services to address unmet customer needs, today Cook Medical announced a strategic investment in the urology space. Zenflow is a medical device company developing a minimally invasive treatment for urinary obstruction caused by enlarged prostate, or benign prostatic hyperplasia (BPH).
“This investment reflects our confidence in the future of Zenflow and the Spring® System technology. Zenflow is aligned with our core focus of developing minimally invasive technologies that restore flow,” said DJ Sirota, senior vice president of Cook Medical’s MedSurg division. “This type of agreement is yet another proof-point to how Cook is changing to focus on our future as product innovators.”
With funding led by Cook, Zenflow closed an $24 million Series C financing round today and included participation from existing investors Invus Opportunities, F-Prime Capital, Medical Technology Venture Partners and others.
Funds will be used to support the company’s Pre-Market Approval (PMA) submission as well as to prepare for commercialization in order to set the stage for FDA approval of its Spring System, a minimally invasive treatment option for patients who suffer debilitating symptoms due to enlarged prostate. The Spring System was designed with the patient’s experience in mind and relies on a small spring-like coil to gently prop open the urethra, restoring its normal function while preserving the natural anatomy. Over 40 million patients suffer from BPH in the U.S.







